Cell design refers to a method of assembling positive electrode material, negative electrode material, electrolyte, separator, and current collectors in certain proportions and processes to meet specific electrical performance requirements. The designer must understand the characteristics of each component, including the positive electrode material, negative electrode material, electrolyte, separator, and conductive agents, and possess extensive knowledge of electrochemistry, as well as a holistic approach to thinking. This is a massive manufacturing project that involves various processes, each with its own technical characteristics.
Clear Design Objectives
Firstly, it is essential to clearly define the requirements that the designed cell must meet. Currently, the demand for cells mainly comes from two aspects: one is the internal technological storage of battery companies, anticipating market needs in advance; the other is from enterprises that require cells, whether consumer batteries or power batteries, which necessitates translating the language of corporate needs into the language of cells, as follows:
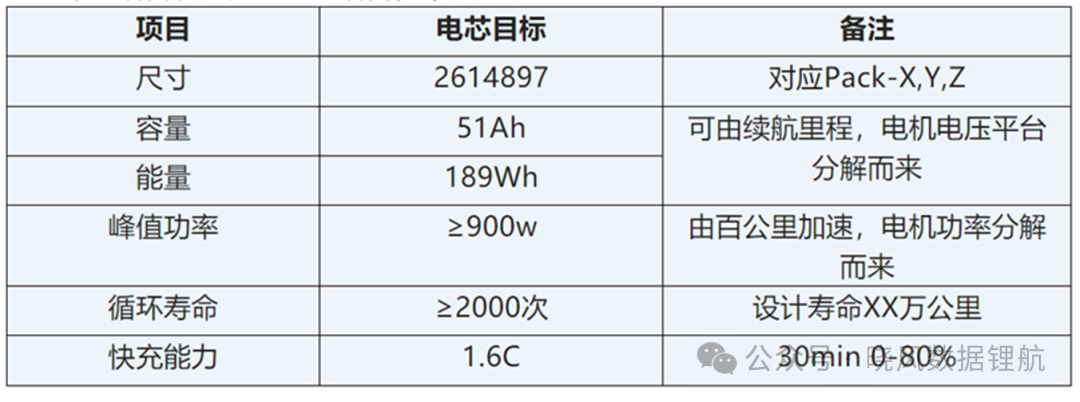
Design Fundamentals
The basic principle of cell design is to find a relative balance among parameters such as energy density, lifespan, rate capability, and cost, as it is difficult to satisfy all indicators simultaneously. Therefore, it is very important to classify the objectives according to customer needs. Classify based on the following: (1) Customer's essential needs must be met, such as capacity, energy, and safety; (2) Customer's special requirements should be met as much as possible, such as fast charging and power output; (3) Non-essential customer requirements. To identify these, multiple and in-depth communications with customers are needed to avoid Design deviation
Selection of Chemical Systems (Taking lithium batteries as an example)
Selection of the cathode: The choice of cathode material determines the energy density of the cell. NMC (Nickel-Manganese-Cobalt Oxide) and LFP (Lithium Iron Phosphate) are commonly used cathode materials for power cells, while LCO (Lithium Cobalt Oxide) is more commonly used for consumer applications. Whether for consumer or power cells, the basic design concept is essentially the same. Taking square power cells as an example, if our design goal is to achieve 210 Wh/kg, then normally 5-series or higher NMC materials are required; to achieve 240 Wh/kg or more, 8-series or higher NMC materials are needed. Currently, there is a division in the industry over the use of 8-series materials. Therefore, when choosing cathode materials, a different approach is needed. We all know that energy density is mainly determined by capacity (C) and voltage (V). When high-capacity materials are not an option, we must increase the material's cut-off voltage, such as from 4.2 V to 4.35 V. In addition, if increasing capacity and voltage still cannot fully meet the energy density requirements, further optimization of the cell structure and process is needed.
Selection of the anode: Anode materials are often artificial graphite, which can also be doped with silicon, or use lithium titanate, hard carbon, etc. For artificial graphite paired with NMC materials, its specific capacity is generally already around 350 mAh/g. For a given case, increasing the specific capacity of the anode can reduce the amount of anode material needed, thereby increasing the energy density of the cell. Doping graphite anodes with silicon is a relatively effective method, but silicon doping can lead to increased anode expansion, reduced initial efficiency, and challenges to fast charging capability and cycle life. This is its drawback. Therefore, when deciding to add silicon, the amount of doping and subsequent lithium compensation process should be considered simultaneously.
Selection of the Diaphragm: When choosing a diaphragm, not only its material, thickness, and cost need to be considered, but also its porosity, air permeability, ceramic coating weight, and whether it is coated with PVDF. Ceramic coating can improve the safety performance of the cell, while coating with PVDF can optimize the adhesion of the cell interface. However, this also brings about an increase in process complexity and cost, which designers need to carefully consider based on their requirements.
Selection of the Electrolyte: The electrolyte plays a role in ion conduction and electronic insulation between the positive and negative electrodes of a battery. In secondary lithium batteries, the properties of the electrolyte have a significant impact on the battery's cycle life, operating temperature range, charge-discharge efficiency, safety, and power density. Secondary lithium battery electrolyte materials should possess the following properties: (1) high lithium-ion conductivity; (2) high electrochemical stability, maintaining temperature over a wide potential range; (3) good compatibility with electrodes, effectively forming a stable SEI film on the negative electrode and having sufficient anti-oxidation decomposition ability at high potentials on the positive electrode; (4) good contact with electrodes, for liquid electrolytes, fully wetting the electrodes; (5) good low-temperature performance, maintaining high conductivity and low viscosity in a lower temperature range (-20~20 ℃) to ensure good electrode surface wettability during charge-discharge processes; (6) a wide voltage range; (7) good thermal stability, not undergoing thermal decomposition over a wide temperature range; (8) good chemical stability, not undergoing chemical reactions during long-term cycling and storage of the battery, and not reacting with the positive electrode, negative electrode, current collector, binder, conductive agent, diaphragm, packaging material, or sealant; (9) non-toxic, non-polluting, safe to use, and preferably biodegradable; (10) easy preparation and low cost. Common electrolyte additives include SO2/CO2/VC (vinylene carbonate) [to improve SEI film performance], trimethyl phosphate (TMP) [to improve electrolyte safety], crown ethers [to increase electrolyte conductivity], Al2O3, MgO, and carbonates of lithium or calcium [to control water and acid content].
Therefore, it is necessary to select an appropriate electrolyte based on the performance that the cell can achieve.
Selection of Current Collectors: The current collector is one of the indispensable components in lithium-ion batteries. It not only carries the active materials but also aggregates and outputs the current generated by the electrode active materials, which helps to reduce the internal resistance of lithium-ion batteries, improve the coulombic efficiency, cycling stability, and rate performance. Ideally, the current collector for lithium-ion batteries should meet the following conditions: (1) High electrical conductivity; (2) Good chemical and electrochemical stability; (3) High mechanical strength; (4) Good compatibility and adhesion with electrode active materials; (5) Affordable and readily available; (6) Light weight. However, in practical applications, different current collector materials still have various issues, and thus cannot fully meet the above multi-scale requirements. For instance, copper is prone to oxidation at higher potentials and is suitable for use as a negative electrode current collector; whereas aluminum has more severe corrosion issues when used as a negative electrode current collector and is more suitable for use as a positive electrode current collector. Currently, materials that can be used as current collectors for lithium-ion batteries include metal conductors such as copper, aluminum, nickel, and stainless steel, semiconductor materials such as carbon, and composite materials.
Copper is an excellent metal conductor with electrical conductivity second only to silver, possessing numerous advantages such as abundant resources, affordability, and good malleability. However, considering that copper is prone to oxidation at higher potentials, it is often used as the current collector for negative active materials such as graphite, silicon, tin, and cobalt-tin alloys. Common copper current collectors include copper foil, copper foam, copper mesh, and three-dimensional nano-copper array current collectors.
(1) Copper foil current collector. According to the production process of copper foil, it can be further divided into rolled copper foil and electrolytic copper foil. Compared with electrolytic copper foil, rolled copper foil has higher electrical conductivity and better elongation effects. Lithium-ion batteries that do not require high bending tolerance can choose electrolytic copper foil as the negative current collector. Studies have shown that increasing the roughness of the copper foil surface is beneficial for enhancing the bonding strength between the current collector and the active material, reducing the contact resistance between the active material and the current collector, and correspondingly, the battery's rate discharge performance and cycle stability are also improved.

(2) Copper foam current collector, copper foam is a three-dimensional network material similar to a sponge, possessing many advantages such as light weight, high strength and toughness, and large specific surface area. Although silicon and tin anode active materials have high theoretical specific capacities and are considered to be one of the most promising anode active materials for lithium-ion batteries, they also have drawbacks such as significant volume changes and pulverization during the charge/discharge cycling process, which severely affect battery performance. Studies have shown that the copper foam current collector can suppress the volume changes of silicon and tin anode active materials during the charge/discharge process, slow down their pulverization phenomenon, and thus improve battery performance.

Although the conductivity of aluminum is lower than that of copper, aluminum wires only need half the mass of copper wires to carry the same amount of electricity. Undoubtedly, using aluminum current collectors helps to increase the energy density of lithium-ion batteries. Additionally, aluminum is more cost-effective compared to copper. During the charging and discharging process of lithium-ion batteries, a dense layer of oxide film forms on the surface of the aluminum foil current collector, enhancing its corrosion resistance. Aluminum foil is commonly used as the current collector for the positive electrode in lithium-ion batteries. Like copper foil current collectors, surface treatment can also improve the surface characteristics of aluminum foil. After direct current etching, a honeycomb-like structure forms on the surface of the aluminum foil, which binds more closely with the positive electrode active material and improves the electrochemical performance of lithium-ion batteries. However, in reality, aluminum current collectors often suffer from severe corrosion due to the damage of the surface passive film, which in turn reduces the performance of lithium-ion batteries. Therefore, to enhance the corrosion resistance of etched aluminum foil, it is necessary to optimize its surface treatment to form a more stable passive film.

Relatively speaking, nickel is a base metal with a lower price, good electrical conductivity, and stability in both acidic and alkaline solutions. Therefore, nickel can serve as both a positive and negative current collector. It pairs well with positive active materials such as lithium iron phosphate and negative active materials like nickel oxide, sulfur, and carbon-silicon composites. Nickel current collectors typically come in two forms: foam nickel and nickel foil. Due to the developed pore structure of foam nickel, it has a large contact area with the active material, thereby reducing the contact resistance between the active material and the current collector. However, when nickel foil is used as the electrode current collector, the active material tends to fall off with increasing charge/discharge cycles, affecting the battery performance. Surface pretreatment processes are also applicable to nickel foil current collectors. For instance, after etching the surface of the nickel foil current collector, the bonding strength between the active material and the current collector significantly increases. Nickel oxide has advantages such as stable structure, low cost, and a high theoretical specific capacity, making it a widely used negative active material for lithium-ion batteries. Based on this, a nickel oxide negative electrode can be prepared by in-situ growth of nickel oxide on the surface of foam nickel using the solid-phase oxidation method. Compared to nickel foil/nickel oxide negative electrodes, the initial discharge specific capacity of foam nickel/nickel oxide negative electrodes is significantly increased. The reason is that compared to two-dimensional current collectors, three-dimensional current collectors reduce interfacial polarization phenomena and improve the charge/discharge cycle stability of the battery. Lithium iron phosphate is considered an ideal positive active material for power lithium-ion batteries due to its good safety and wide availability of raw materials. Coating it on the surface of foam nickel current collectors can increase the contact area between LiFePO4 and foam nickel, reduce the current density of the interfacial reaction, and thereby improve the rate discharge performance of LiFePO4.
Stainless steel refers to alloy steel containing elements such as nickel, molybdenum, titanium, niobium, copper, and iron. It has good electrical conductivity and stability and can withstand chemical corrosion from weak corrosive media such as air, steam, water, and strong corrosive media such as acids, bases, and salts. Stainless steel surfaces can also easily form a passive film that protects them from corrosion. Additionally, stainless steel can be processed thinner than copper, offering advantages such as low cost, simple processes, and the ability to produce in large quantities. Stainless steel can serve as a current collector for either the positive or negative electrode, with common types being stainless steel mesh and porous stainless steel.
(1) Stainless steel mesh current collector, due to its dense texture, when used as a current collector, its surface is wrapped by the electrode active material and does not directly contact the electrolyte, reducing the likelihood of side reactions and helping to improve the battery's cycle performance.
(2) Porous stainless steel current collector, to fully utilize the active material and increase the electrode's discharge specific capacity, a simple and effective method is to use a porous current collector.
Using carbon materials as positive or negative current collectors can avoid corrosion of the metal current collector by the electrolyte and has advantages such as abundant resources, ease of processing, low resistivity, no environmental harm, and low cost. Carbon fiber cloth, with its inherent flexibility, conductivity, and electrochemical stability, can be used as a current collector for flexible lithium-ion batteries. Carbon nanotubes are another form of carbon current collector. Compared to metal current collectors, their significant advantage is their light weight, which can greatly increase the energy density of the battery.
In addition to single current collectors such as copper, aluminum, nickel, stainless steel, and carbon current collectors that have received widespread attention, composite current collectors have also sparked the interest of researchers in recent years, examples include conductive resins, carbon-coated aluminum foil, and titanium-nickel shape memory alloys.
Polyethylene (PE) and phenolic resin (PF) current collectors are formed by compounding conductive fillers with polymer resin matrices. Using PE and PF as the base materials, they are uniformly mixed with conductive fillers (graphite, carbon black) to prepare composite current collectors, and their physical and chemical properties are studied. Graphene is a unique new two-dimensional carbon functional material formed by sp2 hybridization of carbon atoms, possessing numerous advantages such as ultra-high electrical conductivity, specific surface area, and mechanical strength. It can replace graphite as the anode active material for lithium-ion batteries and can also serve as a current collector material.
Titanium-nickel shape memory alloy is a binary alloy composed of nickel and titanium, which can transform between two different crystal phases with changes in external temperature or pressure. Titanium-nickel shape memory alloy can suppress the volume change of active materials during the charge-discharge process by changing its phase state, thereby improving the cycle life of the battery.
A carbon-coated/aluminum foil current collector is a composite current collector that involves applying a carbon composite layer onto the surface of aluminum foil. The carbon layer consists of carbon fibers and conductive carbon black particles treated with a dispersant, which can bond tightly with the aluminum foil, enhancing the conductivity and corrosion resistance of the electrode.

The current collector is one of the indispensable and important components in lithium-ion batteries, possessing multiple functions such as carrying the electrode active materials and collecting and outputting the current. The performance of current collectors prepared from different materials and production processes varies greatly, and their impact on lithium-ion batteries is also different.
Capacity Design
The simple calculation formula for cell capacity is as follows:
Design Capacity = Cathode Material's Gram Capacity * Coating Weight * Loading * Electrode Length * Electrode Width * 2 * Core Number
The cathode material's gram capacity is provided by the material manufacturer when the product is released, and of course, the cell manufacturer will also test it to ensure the stability of the batch, which can also improve the consistency of subsequent pairing, and the test method is usually a button cell with a lithium electrode. The coating weight is measured in g/m², and the coating methods are transfer and extrusion coating. Since extrusion coating has advantages in tolerance and precision, most mass production lines currently use extrusion coating. Loading refers to the mass percentage of the cathode active material in the cathode formulation. Most mass-produced square cell cores have a number of 2 or 4. The possible reason why fewer manufacturers achieve 1 is that from a process perspective, the electrode is too long, which can lead to easy breakage during die cutting, affecting the yield; too many layers can also make it difficult to control the misalignment during winding.
N/P Design
N/P = Negative electrode capacity * Negative active material mass / Positive electrode capacity * Positive active material mass. The main purpose is to ensure that at the same time and location, the lithium intercalation capability of the negative electrode is greater than the lithium deintercalation capability of the positive electrode. The capacity per gram is usually obtained from charge-discharge tests, hence there are typically two N/P values: one for the first charge and another for the second discharge. The design of the first charge N/P is mainly to ensure that no lithium plating occurs during the formation stage, while the design of the second discharge N/P is to ensure long cycle life in subsequent use. Therefore, the actual selection of the N/P value must take into account both charging and discharging to make the optimal choice.
Safety Design
When designing the internal structure of the cell, safety is a key consideration. Common strategies include:
Overhang Design: The length and width of the separator should be able to cover the negative electrode, and the length and width of the negative electrode should be able to cover the positive electrode. This approach has several benefits: first, it can prevent internal short circuits caused by the expansion of the electrode plates during charge and discharge; second, it can prevent internal short circuits caused by the compression of the separator.
Coating Ceramic on Positive Electrode Edges: This involves applying an insulating ceramic layer to the edges of the positive electrode to prevent short circuits caused by the positive and negative electrodes touching.
Top Cap Safety Design: This mainly includes fuse melting design and overcharge protection (OSD) flip design. The design must consider the overcurrent capacity of the fuse, and relevant validation data should be collected and tested in advance for both sustained overcurrent and peak overcurrent. The OSD is mainly to prevent overcharging, and an earlier article has introduced its mechanism. With the release of new national standards, the NCM system may pass the overcharge test without OSD. Therefore, to improve the reliability of the top cap and reduce costs, OSD may gradually be phased out in the future.
Positive and Negative Electrode Formulation Design
The positive electrode formulation typically consists of ternary materials, conductive agents, binders, etc., with NMP as the solvent. Common conductive agents include SP, KS-6, and CNT, and binders are commonly PVDF. To maximize the energy density of the cell, the loading of NCM should be at least 95% or more. The reason loading cannot reach 100% is that the role of auxiliary materials is also very important. The functions of conductive agents and binders are self-explanatory from their names, so they will not be explained in detail here. It is worth noting that SP is a chain-like conductive agent that serves a long-range conduction function, while KS-6 is a flake-like material that serves a short-range conduction function. In practical applications, both must be used in combination to better enhance the conductivity.
The formulation for the negative electrode is similar, mainly consisting of graphite/silicon, conductive agents, binders, emulsifiers, etc. The content of the main material should also be maximized, generally above 96%. The type of conductive agent is the same as that of the positive electrode, but the binders and emulsifiers are different. Common binders include SBR (styrene-butadiene rubber) and polyacrylate materials, which not only enhance the bonding ability but also accelerate the transmission of lithium ions. Currently, the solvent for graphite negative electrode slurries is mostly deionized water. Since graphite is a non-polar substance with low surface energy, and the solvent water is polar, the hydrophobic bonds of the emulsifier CMC-Na (sodium carboxymethyl cellulose) adsorb onto the graphite surface through van der Waals forces, while the hydrophilic groups combine with water to enhance the hydrophilicity of graphite. At the same time, after being coated with CMC-Na, the negative charge on the graphite surface increases, causing greater repulsion between particles, making the slurry less prone to sedimentation. Therefore, CMC-Na plays a role in enhancing the hydrophilicity of graphite and providing steric hindrance.
Source: Xiji Battery and Energy Storage WeChat Public Account
https://mp.weixin.qq.com/s/TZzn6S9BngnTw0id9EmMYQ
 Analysis and Demand Forecast of Consumer Lithium Battery Market in 2024!March 31, 2026Part 1: Steady Growth in Traditional Consumer Electronics1.1 Smartphones: The global shipment growth rate is expected to be 4% in 2024. According to IDC, the global smartphone shipment growth rate is ...view
Analysis and Demand Forecast of Consumer Lithium Battery Market in 2024!March 31, 2026Part 1: Steady Growth in Traditional Consumer Electronics1.1 Smartphones: The global shipment growth rate is expected to be 4% in 2024. According to IDC, the global smartphone shipment growth rate is ...view Understanding the Impact of High and Low Temperatures on Lithium Battery PerformanceMarch 31, 2026Lithium iron phosphate batteries and ternary lithium batteries in lithium batteries have the advantages of high energy density, wide operating temperature range, long cycle life, and safety and reliab...view
Understanding the Impact of High and Low Temperatures on Lithium Battery PerformanceMarch 31, 2026Lithium iron phosphate batteries and ternary lithium batteries in lithium batteries have the advantages of high energy density, wide operating temperature range, long cycle life, and safety and reliab...view The Qingdao Energy Institute Has Published a Review on the Breakthrough in Regulating Multi-level Structures to Overcome the Charge Transmission Bottleneck in Solid-State Lithium Batteries.January 21, 2026Solid-state lithium metal batteries are regarded as the ideal choice for next-generation energy storage technologies due to their high energy density and excellent safety. However, the slow charge tra...view
The Qingdao Energy Institute Has Published a Review on the Breakthrough in Regulating Multi-level Structures to Overcome the Charge Transmission Bottleneck in Solid-State Lithium Batteries.January 21, 2026Solid-state lithium metal batteries are regarded as the ideal choice for next-generation energy storage technologies due to their high energy density and excellent safety. However, the slow charge tra...view Beyond Traditional Power: How JREPower Lithium Batteries Reshape Floor Cleaning EfficiencyFebruary 3, 2026Is your floor cleaning machine still compromising on runtime and efficiency?Today, floor cleaning machines in commercial and industrial fields widely rely on traditional lead-acid batteries. These wer...view
Beyond Traditional Power: How JREPower Lithium Batteries Reshape Floor Cleaning EfficiencyFebruary 3, 2026Is your floor cleaning machine still compromising on runtime and efficiency?Today, floor cleaning machines in commercial and industrial fields widely rely on traditional lead-acid batteries. These wer...view Basic knowledge of lithium batteries......March 31, 2026Introduction to Lithium MetalLithium is a metallic element that sits at the top of the metal category in the periodic table. Its chemical symbol is Li, and it is a silvery-white, very soft metal with ...view
Basic knowledge of lithium batteries......March 31, 2026Introduction to Lithium MetalLithium is a metallic element that sits at the top of the metal category in the periodic table. Its chemical symbol is Li, and it is a silvery-white, very soft metal with ...view Mechanism of SEI Film Formation in Lithium-Ion Batteries and the Impact of Formation Processes!March 31, 2026In the production process of lithium-ion batteries, formation is an extremely important step. This step mainly includes the initial charging process of the cell after the electrolyte injection is comp...view
Mechanism of SEI Film Formation in Lithium-Ion Batteries and the Impact of Formation Processes!March 31, 2026In the production process of lithium-ion batteries, formation is an extremely important step. This step mainly includes the initial charging process of the cell after the electrolyte injection is comp...view