In the production process of lithium-ion batteries, formation is an extremely important step. This step mainly includes the initial charging process of the cell after the electrolyte injection is completed, which activates the active materials in the battery, making the lithium-ion battery activated; at the same time, the solvent of the electrolyte and the lithium salt undergo side reactions, forming a solid electrolyte interphase (SEI) film on the negative electrode of the lithium-ion battery.

This film will prevent further occurrence of side reactions, thereby reducing the loss of lithium content in lithium-ion batteries. Therefore, it has a significant impact on the initial capacity loss, cycle life, rate performance, and safety of lithium-ion batteries.
1.1 Formation Mechanism of SEI Film
During the formation of the SEI film, various reduction reactions occur involving the electrolyte solvent, lithium salt, additives, trace air impurities, and more. This series of reactions is influenced by both inherent properties of the materials, such as reduction potential, reduction activation energy, and exchange current density, and by other factors such as temperature, electrolyte salt concentration, and reduction current. The combined effects of these factors complicate the formation process of the SEI film, making the mechanism difficult to understand clearly.
It is generally accepted that the formation of the SEI film occurs in two stages: first, the negative electrode of the battery polarizes, and the components of the organic electrolyte solution undergo reduction decomposition, forming new chemical products; subsequently, the newly formed products precipitate on the negative electrode surface to form the SEI film. In the study of the SEI film formation mechanism, the controversy mainly focuses on the process of reduction reactions, especially the reduction reaction process of electrolyte solvent molecules. These reduction reaction processes are inferred from the battery negative electrode polarization, thermodynamic simulations, ion size, and other factors indicating the transfer of electrons in the electrolyte solution towards the salt and solvent.
There are three possible scenarios in this process: First, during the reduction reaction, individual lithium ions directly intercalate into the negative electrode graphite layer, completing the intercalation; second, according to the hypothesis of Aurbach et al., the reduction reaction of solvent molecules is a single-electron reaction, generating an intermediate product—a free radical anion. This anion further decomposes and combines with lithium ions to form a precipitate, becoming a component of the SEI film. According to the hypotheses of Dey, Besenhard, and Chung, when lithium ions and solvent molecules co-intercalate, the solvent molecules undergo a two-electron reduction reaction, binding with lithium ions through weak van der Waals forces to co-intercalate into the graphite layer, forming an intermediate product—ternary graphite intercalation compounds. This intermediate product is then reduced to form the SEI film; third, electrons from the negative electrode directly transfer to the anion salt, combining with lithium ions to directly form an inorganic salt precipitate.
1.2 Structure and Properties of the SEI Film
During the practical formation process of lithium-ion batteries, due to the complexity of chemical reactions, the presence of numerous impurities, and uneven current distribution, the structure of the SEI film is quite complex. The mainstream viewpoint holds that it is a bilayer structure: the side close to the electrolyte is porous and loose, mostly composed of organic compounds, and the pores of this layer are filled with electrolyte. This layer's structure may undergo further reduction in subsequent cycles, changing its morphology; the side close to the anode is mainly composed of inorganic compounds, with fewer pores and a compact structure.
The SEI film is a fragile thin layer structure. A fully formed SEI film has a high lithium-ion conductivity and negligible electronic conductivity, sufficient flexibility, and is robust enough. The electronic insulating properties of the SEI film prevent further reduction reactions of the electrolyte on the anode surface; the ionic conductivity allows lithium ions to pass through the SEI film and intercalate into the anode; the robustness and flexibility of the SEI film can prevent the volume changes of the anode material during lithium-ion deintercalation from causing the SEI film to rupture. At the same time, there is a sufficiently strong molecular force between the SEI film and the anode surface, which can prevent subsequent further polarization reactions.
Due to the numerous reactants involved in the formation of the SEI film, and the unstable composition of the electrolyte, along with varying reaction conditions, the products of the reduction reactions are diverse. Different research groups have measured different components of the SEI film, but overall, there are some common patterns. For example: when fluorinated salts such as LiAsF6, LiPF6, LiBF4 are present in the electrolyte, they undergo reduction reactions and precipitate as LiF or LixPFy; carbonates in the electrolyte react with lithium salts to precipitate as Li2CO3, ROCO2Li, or other organic compounds; after a two-electron reduction reaction of ethylene carbonate in the electrolyte, (CH2OCO2Li)2 precipitates appear on the SEI film; when the content of propylene carbonate in the electrolyte is high, ROCO2Li precipitates appear on the outer layer of the SEI film, and so on.
1.3 SEI Membrane Characterization
Characterizing the SEI membrane to obtain its various physical and chemical properties is a prerequisite for further analysis of the SEI membrane. However, the SEI membrane is extremely unstable; when exposed to air, its components can easily react with CO2 and H2O in the air to form inorganic lithium salts such as Li2CO3 and Li2O. At the same time, the lithium in the SEI reacts with oxygen to form various nucleophilic oxides, which then react with organic molecules and semicarbonate to produce carbonates and alkoxides. Therefore, when characterizing the SEI membrane, special containers should be used to quickly transfer the sample from an inert gas-filled glovebox to the analytical instrument to avoid chemical contamination and physical damage.
As characterization methods continue to increase, the methods for characterizing the SEI membrane have become diverse. Scanning electron microscopy (SEM), transmission electron microscopy (TEM), atomic force microscopy (AFM), and ellipsometry can be used to obtain images of the surface morphology and characteristics of the SEI membrane. In traditional electrochemical characterization methods, electrochemical impedance spectroscopy (EIS) and cyclic voltammetry (CV) are commonly used. EIS provides useful information about complex electrochemical systems with diffusion layers, electrolyte resistance, electrode kinetics, and double-layer capacitance by establishing equivalent circuit models; CV can measure currents in the anodic and cathodic directions, which helps to better understand the SEI membrane. Due to the SEI membrane's extreme thinness, X-ray photoelectron spectroscopy (XPS) and FTIR, which have high surface sensitivity and chemical identification capabilities, can be used for surface analysis, while Raman and X-ray diffraction (XRD) can be used to identify the types of substances on the SEI membrane surface.
1.4 Effect of Electrolyte on SEI Film
The electrolyte is the main source of reactants for the reduction reaction that forms the SEI film, and both the electrolyte solvent and lithium salts affect the composition of the SEI film. Once the main components of the electrolyte are fixed, performance of the SEI film can only be improved through other means.
Researchers have found that the formation of the SEI film begins with electrons transferring from the cathode-polarized electrode to solvated Li+, followed by the establishment of a charge exchange equilibrium between the solvated Li+ complexed solvent molecules and the solvent molecule radicals generated. During this process, the soluble reduction compounds that are formed are re-oxidized, and charge transfer requires an empty molecular orbital from the electrode to the solvent molecules complexed with ions. When this empty orbital has relatively high energy, charge transfer can only occur at a more negative potential, meaning that substances reduced at higher potentials are the most reactive.
Therefore, people consider adding substances with higher activity to the electrolyte to enhance the performance of the SEI film, namely film-forming additives. These additives have active groups with strong electron-absorbing capabilities, which can increase the reduction potential and undergo reduction reactions before lithium ions intercalate into the graphite anode, forming a passivation film. This suppresses further decomposition of the electrolyte and improves the performance of the SEI film.
Researchers divide film-forming additives into organic compound additives and inorganic compound additives. Organic compound additives are the main component of film-forming additives, among which unsaturated carbon compounds such as vinylene carbonate (VC) have a double bond structure that makes them easier to reduce due to their lower energy. Propylene sulfite (PS) and ethylene sulfite (ES) can also improve the performance of lithium-ion batteries. Organic compound additives containing halogens also significantly help enhance the performance of lithium-ion batteries. Adding fluoroethylene carbonate (FEC) to silicon anode lithium-ion batteries has been found to promote the formation of LiF and polycarbonate compounds, reduce the impedance of the SEI film on the silicon surface, and thus improve the cycling performance of lithium-ion batteries.
When studying inorganic compound additives, it was discovered that CO2, SO2, CS2, Sx2-, N2O, and other gases can react with the electrolyte to form Li2CO3, Li2S, Li2SO4, and Li2O, which can also improve the electrochemical performance of lithium-ion batteries. However, these gaseous compounds are difficult to dissolve in organic solvents, which is not conducive to production practice, so inorganic salts such as Li2CO3, K2CO3, NaClO4, and AgPF6 are used as substitutes. Compared to organic compound additives, inorganic compound additives are non-flammable, which increases the safety of lithium-ion batteries.
1.5 Effect of SEI Film on Lithium-Ion Battery Performance
Extensive research has found that the properties of the SEI film greatly affect the performance of lithium-ion batteries. During the formation process, the amount of SEI film formed represents the amount of lithium consumed in the lithium-ion battery, directly determining its capacity. Therefore, in the process of forming the SEI film, the less lithium consumed, the better, meaning that the smaller the irreversible capacity loss, the better.
During the cycling of lithium-ion batteries, if the electronic insulation properties of the SEI film are poor, electrons will contact the electrolyte, and the reduction reaction will continue, consuming the lithium content in the battery and causing the continuous formation of the SEI film, resulting in poor cycling life of the lithium-ion battery.
During the cycling process, the SEI film may experience two phenomena: peeling and thickening. When peeling occurs, the resulting SEI film fragments enter the electrolyte and undergo electrophoresis under voltage, especially during high-rate discharge, where the resulting fragments deposit on the electrode surface. At the same time, during high-rate cycling, the SEI film on the negative electrode of the lithium-ion battery will significantly thicken. Both phenomena increase the surface resistance of the electrode, affecting the deintercalation of lithium ions, and thus affecting the rate performance of the lithium-ion battery.
During fast charging of lithium-ion batteries, if the speed of lithium ions passing through the SEI film is slower than the speed of lithium deposition on the negative electrode, lithium dendrites may continuously form with charge-discharge cycles. This could lead to short circuits in the lithium-ion battery, causing combustion and explosion. Additionally, when the SEI film is incompletely formed or decomposes, the lithium intercalated into the negative electrode will react with the electrolyte and binder, releasing heat. The reaction heat increases with the amount of lithium intercalation, greatly affecting the safety of the battery. The SEI film has a direct or indirect impact on battery performance, and forming an SEI film that meets requirements greatly helps to improve battery performance.
2.1 Overview of the Formation Process
In the formation process of lithium-ion batteries, charging methods include constant current, constant voltage, and intelligent charging. Constant current charging has severe polarization, a lower initial current, a higher final current, a longer charging time, significant energy waste, and can reduce battery life. During constant voltage charging, the initial stage has a larger current, and the electromotive force of the battery gradually increases until the charging current gradually drops to zero. Compared to the former, constant voltage charging consumes less energy and has a shorter charging time, with the charging performance being closer to the optimal charging curve; however, it is difficult to compensate for the changes in battery terminal voltage during the charging process, and improper selection of charging voltage can damage the battery.
Intelligent charging dynamically tracks the charging current that the battery can accept during the charging process. The charging power supply can automatically adjust the charging parameters according to the state of the battery, maintaining the charging current within an allowable range to protect the battery. However, it is difficult to dynamically track the charging current that the battery can accept in actual situations. Therefore, in practical applications, the constant current and constant voltage charging method is more commonly used, which is to use constant current charging in the initial stage, and when the battery terminal voltage rises to a certain value, switch to constant voltage charging until the battery is fully charged.
The discharge method for batteries is relatively simple, usually constant current discharge, with the voltage gradually decreasing until it reaches the set voltage. Since the SEI (Solid Electrolyte Interface) film begins to form during the charging stage, and most of the SEI film is generated during the first charge-discharge cycle, the parameter control during this stage plays a decisive role in the performance of the generated SEI film. The three most important parameters are: charging current, formation temperature, and cut-off voltage.
2.2 Effect of Formation Current Density on Battery Performance
When graphite is used as the negative active material, there are two types of reactions that form the SEI film: a double-electron reaction, which requires two electrons to participate in the reaction, making it easier to generate inorganic lithium salt components; and a single-electron reaction, which requires only one electron to participate, making it easier to generate organic lithium salt components.
In the initial stage of SEI film formation, a large number of electrons accumulate on the surface of graphite particles, making it easier to undergo a double-electron reaction with film-forming additives and lithium ions, thus forming an SEI film that is primarily composed of inorganic lithium salts; in the later stages of film formation, electrons need to pass through the already formed SEI film to react with film-forming additives and lithium ions, reducing the number of electrons that reach the reaction point, making it easier to undergo a single-electron reaction, and thus forming an SEI film that is primarily composed of organic lithium salts. Therefore, under different charging currents, the composition and structure of the SEI film are different.
By using EIS (Electrochemical Impedance Spectroscopy), in-situ FTIR (Fourier Transform Infrared Spectroscopy), and TEM (Transmission Electron Microscopy) to characterize half-cells at two different current densities of 0.312 and 1.248 μA/cm², it was found that at room temperature, the formation current density chemically affects the formation of the SEI film on the carbon negative electrode: at low current density, Li2CO3 is generated in the early stages of discharge, and alkyl carbonate lithium salts are generated at the end of discharge.
2.3 Effect of Formation Temperature on Battery Performance
On one hand, the formation temperature affects the reaction rate of the chemical reactions that form the SEI film and the corresponding products; on the other hand, as the temperature increases, some components of the SEI film decompose, causing the SEI film to crack and further consume lithium reserves to form a new SEI film.
Lee et al. found that during the formation of the SEI film, EC directly produces ROCO2Li through reduction reactions, which then converts into Li2CO3 while generating gas. The higher the temperature, the more intense this process becomes, producing more gas, and consequently, more defect points are formed on the SEI film, and the film becomes thicker. This provides more pathways for the co-intercalation of lithium ions and solvated solvent molecules, thus further passivating the SEI film on graphite and increasing the irreversible capacity loss of the battery.
Haruta et al. tested battery cycling performance at different temperatures using linear sweep and cyclic voltammetry and found that after the first cycle at 60°C, the battery's areal capacity decreased by 17%, while at 25°C, it decreased by 40%. Moreover, the measured areal capacity during subsequent cycles was also higher at 60°C. Due to the rapid formation of the SEI film at 60°C, with a smooth and uniform structure and mainly stable Li2CO3 composition, graphite surface damage is reduced, thus improving the areal capacity. Compared to pretreatment at 25°C, pretreatment at 60°C reduced graphite decomposition, and the capacity of the graphite electrode increased by 28%.
2.4 Effect of Formation Cut-off Voltage on Battery Performance
The cut-off voltage generally refers to the voltage at which the battery stops charging during constant current charging, usually the voltage at which the reduction reaction is fully completed. Wenren Hongyan et al. found that as charging progresses, the internal voltage of the battery increases, accompanied by gas production. Once the gas production rate exceeds the exhaust rate of the filling hole, gas will accumulate between the separators inside the battery, leading to uneven contact between the separator and the negative electrode surface. This affects the lithium ion intercalation process on the negative electrode surface, causing uneven distribution of lithium ions during the electrochemical reaction and resulting in the deposition of metallic lithium or lithium compounds on the negative electrode surface. Therefore, appropriately lowering the formation voltage can improve the initial charge-discharge efficiency of the battery, reduce the internal resistance of the battery, and enhance the battery cycling performance. Kim et al. found that the higher the voltage, the more unstable the electrolyte, providing more lithium for reduction reactions, which reduces the lithium inventory of the lithium-ion battery. Lowering the formation voltage can also reduce the formation time, saving electrical costs and improving production efficiency in actual production.
3.1 Overview of Si-based Anode Materials
The theoretical specific capacity of Si is as high as 4200mAh/g, which is 10 times that of graphite's theoretical specific capacity; the lithium insertion potential of Si is low, at 0.37mV Li/Li+; moreover, Si is the second most abundant element on Earth and does not harm the environment. The technology for producing nano-silicon is mature and cost-effective, making Si a material of choice for researchers looking to replace graphite as the primary anode material in the future.
However, during the process of lithium insertion and extraction, the volume of Si expands and contracts, with a change rate reaching up to 400%. The mechanical stress changes generated during this process cause the anode material to collapse, the electrode structure to become unstable, and the SEI film on the anode surface to be unstable and continuously formed, thereby reducing its electrochemical performance. Additionally, Si is a semiconductor with low electrical conductivity, and the expansion and contraction of Si nanoparticles cause them to gradually break away from the electron and particle transport network, further reducing their conductivity. These factors greatly limit the performance of Si's electrochemical properties. How to limit the volume change of Si while taking full advantage of its capacity is the main research direction at present.
3.2 Formation Mechanism of SEI Film on Si-based Anode Materials
Like graphite anode materials, a SEI film is also formed at the solid-liquid interface of Si-based anode materials during the formation process. However, the drastic volume changes of Si during lithium insertion and extraction cause the SEI film to rupture and continuously reform, resulting in poor battery cycling performance and low coulombic efficiency. Therefore, current research focuses on how to limit the volume changes of Si through material composites, thereby utilizing Si's capacity advantage.
Among the many studies, the most involved is the composite of silicon and carbon. In actual production, there are various ways to composite silicon and carbon materials. The ideal structure is to adopt coated and embedded silicon-carbon composite structures to help build a stable SEI film. For example, the core-shell structure formed after coating can buffer the expansion of Si to some extent, making the SEI film more stable and also inhibiting the agglomeration of Si particles. Only by first solving the problem of Si's volume expansion can its capacity characteristics be better utilized, making further research on it as an electrode material meaningful.
3.3 Optimizing the Formation Process
When the capacity of Si as an anode material is well utilized, the application of Si-based materials to actual production becomes a reality. It is of practical significance to study the control of formation process parameters from the perspective of battery production technology, thereby affecting the properties of the SEI film formed, and subsequently enhancing battery performance.
In the laboratory, half-cells were fabricated, and Atomic Force Microscopy (AFM) was used in conjunction with conventional characterization methods and lithium-ion battery performance testing methods to explore the range of single variable parameters for forming an ideal SEI film by individually varying the formation current, formation temperature, and cut-off voltage. Then, within the above optimal range of single variables, two different variables were combined to explore the variable combinations and parameter ranges for forming an ideal SEI film under different conditions.
IV. Conclusion and Outlook
In the research of lithium-ion batteries, most researchers focus on the development and preparation of materials, attempting to improve battery performance through material synthesis and modification. Fewer researchers pay attention to the equally important battery production process. The formation process is a crucial step in the battery from assembly to application. By controlling the current, temperature, and cut-off voltage in the formation process, the formation and properties of the SEI film can be influenced, thereby enhancing battery performance.
Si is considered one of the most promising anode materials, but if the issue of its volume expansion cannot be resolved, there is still a long way to go before the commercial application of Si-based materials. For Si-C composite materials that can better utilize the capacity of Si, it is extremely important to improve battery performance by controlling process parameters. By controlling the three process parameters of the formation process during the production of finished batteries to obtain a high-quality SEI film, further generation of the SEI film during battery cycling can be minimized, reducing the total consumption of lithium in the battery, which can improve the coulombic efficiency, cycle life, and safety performance of the battery.
From the public account "Lithium Battery Frontier"
https://mp.weixin.qq.com/s/Fq_DNbXaGyPfa1VAEv-2hA
 Summary of Lithium Battery Export Data and Market Trends in the First Three QuartersMarch 31, 2026AbstractRecently, the European Union officially announced that it will impose a five-year anti-subsidy tariff on electric vehicles imported from China. Specifically, SAIC Group's products will be ...view
Summary of Lithium Battery Export Data and Market Trends in the First Three QuartersMarch 31, 2026AbstractRecently, the European Union officially announced that it will impose a five-year anti-subsidy tariff on electric vehicles imported from China. Specifically, SAIC Group's products will be ...view Celebrating 2 Years: JREPower Indonesia Production Base AnniversaryJanuary 13, 2026Today, JREPower (Shenzhen JREPower Tech Co., Ltd.) grandly celebrated the 2nd anniversary of its production base establishment in Jakarta, Indonesia. The milestone event gathered local government...view
Celebrating 2 Years: JREPower Indonesia Production Base AnniversaryJanuary 13, 2026Today, JREPower (Shenzhen JREPower Tech Co., Ltd.) grandly celebrated the 2nd anniversary of its production base establishment in Jakarta, Indonesia. The milestone event gathered local government...view The Qingdao Energy Institute Has Published a Review on the Breakthrough in Regulating Multi-level Structures to Overcome the Charge Transmission Bottleneck in Solid-State Lithium Batteries.January 21, 2026Solid-state lithium metal batteries are regarded as the ideal choice for next-generation energy storage technologies due to their high energy density and excellent safety. However, the slow charge tra...view
The Qingdao Energy Institute Has Published a Review on the Breakthrough in Regulating Multi-level Structures to Overcome the Charge Transmission Bottleneck in Solid-State Lithium Batteries.January 21, 2026Solid-state lithium metal batteries are regarded as the ideal choice for next-generation energy storage technologies due to their high energy density and excellent safety. However, the slow charge tra...view Beyond Traditional Power: How JREPower Lithium Batteries Reshape Floor Cleaning EfficiencyFebruary 3, 2026Is your floor cleaning machine still compromising on runtime and efficiency?Today, floor cleaning machines in commercial and industrial fields widely rely on traditional lead-acid batteries. These wer...view
Beyond Traditional Power: How JREPower Lithium Batteries Reshape Floor Cleaning EfficiencyFebruary 3, 2026Is your floor cleaning machine still compromising on runtime and efficiency?Today, floor cleaning machines in commercial and industrial fields widely rely on traditional lead-acid batteries. These wer...view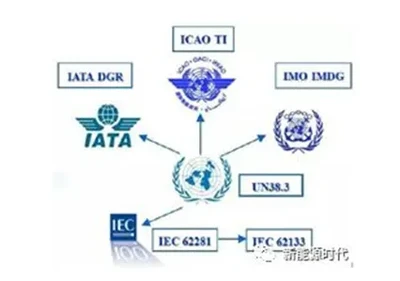 Introduction to Lithium-ion Battery Standards (International Edition)March 31, 2026I. UN38.3UN38.3 is the section of Chapter 38, Paragraph 3 of the United Nations' "Manual of Tests and Criteria for the Transport of Dangerous Goods." In 2003, the United Nations adopted ...view
Introduction to Lithium-ion Battery Standards (International Edition)March 31, 2026I. UN38.3UN38.3 is the section of Chapter 38, Paragraph 3 of the United Nations' "Manual of Tests and Criteria for the Transport of Dangerous Goods." In 2003, the United Nations adopted ...view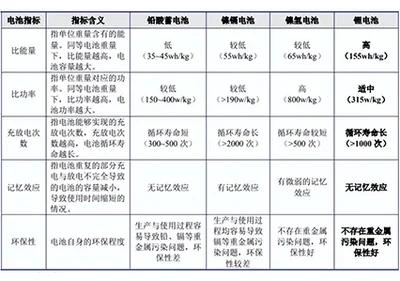 A Guide to Lithium Battery Energy StorageMarch 31, 2026Why are lithium batteries so widely used, what are their advantages, and what are the main applications at present? The following content will sort out the concept definition, working principle, indu...view
A Guide to Lithium Battery Energy StorageMarch 31, 2026Why are lithium batteries so widely used, what are their advantages, and what are the main applications at present? The following content will sort out the concept definition, working principle, indu...view