Lithium is a metallic element that sits at the top of the metal category in the periodic table. Its chemical symbol is Li, and it is a silvery-white, very soft metal with highly reactive chemical properties. It is the lightest metal and is commonly used in the form of compounds as the positive electrode in lithium batteries.
Any chemical power source that uses lithium compounds as the positive electrode is collectively referred to as a lithium-ion battery. Rechargeable lithium-ion batteries are known as secondary batteries (also called storage batteries). They can convert electrical energy into chemical energy for storage and can reverse this process by converting chemical energy back into electrical energy when in use.
Voltage (V): Refers to the potential difference between two points in a circuit, which allows electrons to flow.
Open Circuit Voltage: The voltage of a battery when it is not connected to an external circuit or load. The open circuit voltage is related to the remaining energy of the battery, and the battery level indicator works based on this principle.
Operating Voltage: Also known as terminal voltage, it refers to the potential difference between the positive and negative poles of the battery when it is in operation, i.e., when there is current flowing through the circuit. During the discharge state of the battery, the operating voltage is always lower than the open circuit voltage because the current must overcome the resistance of the internal resistance. The opposite is true during charging.
Discharge Cut-off Voltage: The voltage reached when a fully charged battery is discharged until it is completely empty (if discharged further, it is considered over-discharge, which can damage the battery's life and performance).
Charge Limit Voltage: The voltage value at which the charging process changes from constant current to constant voltage charging.
Internal Resistance (mΩ): The internal resistance of a battery refers to the resistance encountered by the current as it flows through the battery during operation. The size of the internal resistance is mainly influenced by factors such as the battery's material, manufacturing process, and battery structure. The internal resistance of a battery includes both ohmic resistance and polarization resistance. Ohmic resistance is composed of the resistance of the electrode materials, electrolyte, separator resistance, and contact resistance of various components, while polarization resistance includes resistance caused by electrochemical polarization and concentration polarization. Internal resistance is a very complex yet extremely important characteristic of a battery. Factors affecting internal resistance include: ① Main materials; ② Structure. Due to the presence of internal resistance, when the battery discharges, the current passing through the internal resistance generates heat and consumes energy. The greater the current, the more energy is consumed. Therefore, the smaller the internal resistance, the better the battery's performance, not only resulting in a higher actual operating voltage but also less energy lost to the internal resistance.
Battery Capacity (Ah): Battery capacity refers to the amount of electric charge that can be obtained from the battery or the amount of electric charge the battery can store. Capacity is an important indicator of a battery's electrical performance, determined by the capacity of the electrodes. If the capacities of the electrodes are not equal, the battery's capacity is determined by the electrode with the smaller capacity, not the sum of the capacities of the positive and negative electrodes.
Capacity is denoted by C, and the unit is expressed in Ah (Ampere-hours) or mAh (milliampere-hours). The formula is: C=It, meaning Battery Capacity (Ah) = Current (A) x Discharge Time (h).
A battery with a capacity of 10 ampere-hours can discharge for 2 hours at a rate of 5 amperes, or for 1 hour at a rate of 10 amperes. C2 represents the battery capacity measured after discharging completely in 2 hours, and C5 represents the battery capacity measured after discharging completely in 5 hours.
The actual capacity of a battery mainly depends on the following factors: the quantity and quality of the active material; the utilization rate of the active material.
Battery Energy (Wh): Refers to the amount of electricity (energy) stored in the battery, expressed in Wh.
Formula: Energy (Wh) = Rated Voltage (V) × Capacity (Ah).
Significance: The energy of a single cell with 3.7V and 15Ah is 55.5Wh, while a battery pack with 37V and 10Ah has an energy of 370Wh. Battery energy is an important indicator for measuring how much work a battery can drive a device to do; capacity alone cannot determine the amount of work.
Energy Density (Wh/kg): Refers to the energy released per unit volume or unit mass, usually expressed as volumetric energy density (Wh/L) or gravimetric energy density (Wh/kg). For example, a lithium-ion battery cell weighing 325g with a rated voltage of 3.7V and a capacity of 10Ah has an energy density of 113Wh/kg. The values in the table below are theoretical; in practical applications, various factors such as the battery casing and components within the battery structure must be considered. Currently, the energy density of lithium-ion batteries is 3 times that of nickel-cadmium and 1.5 times that of nickel-metal hydride batteries. The level of energy density is determined by the material density and structure.
Discharge Rate (A): The discharge rate refers to the current value required to discharge its rated capacity (C) within a specified time (N), and it numerically equals the multiple of the battery's rated capacity.
The discharge time rate and discharge rate are reciprocals of each other: 1/N rate discharge = N-hour rate discharge; i.e., 1/N * C = C/N.
Using a 10Ah battery as an example:
5-hour discharge rate = C/5 = 10Ah/5h = 2A = 0.2C
0.5-hour discharge rate = C/0.5 = 10Ah/0.5h = 20A = 2C
Charging method: CC/CV: CC stands for Constant Current, which charges the battery with a fixed current; CV stands for Constant Voltage, which charges the battery with a fixed voltage, and the charging current will decrease as the battery approaches full charge.
Trickle charging: Refers to charging the battery with a current less than 0.1C, usually adopted when the battery is nearly fully charged or for supplementary charging. If the load does not have strict requirements for charging time, it is recommended to use trickle charging (in this case, the battery life is longer).
Charge and discharge depth (COD/DOD): A method of expressing the remaining capacity of a battery, charge and discharge depth is represented as a percentage. For example: a 10Ah battery that discharges to 2Ah can be called 80% DOD; a 10Ah battery that charges to 8Ah can be called 80% COD. Describing a fully charged and fully discharged state is commonly referred to as 100% DOD.
State of Charge (SOC): For a 10Ah battery, a state of 5Ah is considered to be 50% SOC.
Self-discharge rate (% per month):
Definition: During the storage (open circuit) process of a battery, the capacity will gradually decrease. The ratio of the reduced electricity to the capacity is called the self-discharge rate.
Cause: Due to the instability of the electrodes in the electrolyte, chemical reactions occur between the two electrodes of the battery, consuming the active materials and reducing the chemical energy that can be converted into electrical energy, leading to a decrease in battery capacity.
Influencing Factors: Environmental temperature has a significant impact on the self-discharge rate, with high temperatures accelerating the self-discharge of the battery.
Representation: The expression method and unit for battery capacity decay (self-discharge rate) is: %/month.
Outcome: Battery self-discharge will directly reduce the battery's capacity, and the self-discharge rate directly affects the battery's storage performance. The lower the self-discharge rate, the better the storage performance.
Cycle Life (cycles):
Concept: A secondary battery undergoes one charge and one discharge as one cycle or one loop. After repeated charging and discharging, the capacity of the battery gradually decreases. Before the electrical capacity drops to a specified value under certain discharge conditions, the number of cycles the battery has undergone is referred to as the cycle life.
Definition: The cycle life is defined as the number of charge-discharge cycles a battery can undergo before its capacity drops to 80% of its rated capacity.
Factors affecting cycle life: Incorrect use of the battery, battery materials, composition and concentration of the electrolyte, charge and discharge rates, depth of discharge (DOD%), temperature, manufacturing processes, and other factors can all impact the cycle life of a battery.
Discharge plateau: refers to the part of the discharge curve where the voltage remains essentially horizontal. The longer and smoother the discharge plateau, the better the battery's discharge performance.
Consistency of Battery Packs: A battery pack is composed of multiple individual cells connected in series and parallel. The overall performance and lifespan of the battery pack depend on the cell with the worst performance, which requires high consistency in the performance of each cell within the battery pack. In addition to the performance errors of the individual cells and the quality of the raw materials, the main reason is the manufacturing process. Improvements in the process are very important for enhancing the quality of the batteries.
Formation: After the battery is made, a certain charging and discharging method is used to activate the internal positive and negative active materials, improving the battery's charge and discharge performance, self-discharge, storage, and other comprehensive performance. This process is called formation. Only after formation can the battery truly reflect its actual performance. At the same time, the sorting process during formation can improve the consistency of the battery pack, thereby enhancing the performance of the final battery pack.
From the public account Lithium Battery Science Society
https://mp.weixin.qq.com/s/2QNzpydqUcGrka_NcCtEIw
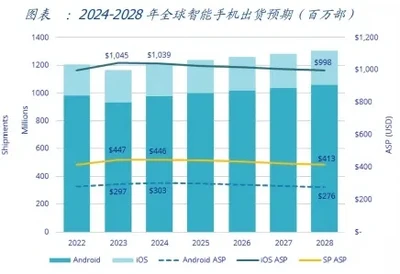 Analysis and Demand Forecast of Consumer Lithium Battery Market in 2024!March 31, 2026Part 1: Steady Growth in Traditional Consumer Electronics1.1 Smartphones: The global shipment growth rate is expected to be 4% in 2024. According to IDC, the global smartphone shipment growth rate is ...view
Analysis and Demand Forecast of Consumer Lithium Battery Market in 2024!March 31, 2026Part 1: Steady Growth in Traditional Consumer Electronics1.1 Smartphones: The global shipment growth rate is expected to be 4% in 2024. According to IDC, the global smartphone shipment growth rate is ...view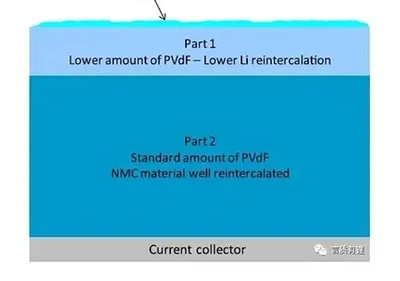 Understanding the Impact of High and Low Temperatures on Lithium Battery PerformanceMarch 31, 2026Lithium iron phosphate batteries and ternary lithium batteries in lithium batteries have the advantages of high energy density, wide operating temperature range, long cycle life, and safety and reliab...view
Understanding the Impact of High and Low Temperatures on Lithium Battery PerformanceMarch 31, 2026Lithium iron phosphate batteries and ternary lithium batteries in lithium batteries have the advantages of high energy density, wide operating temperature range, long cycle life, and safety and reliab...view How are lithium battery cells designed? Which chemical system should be chosen?March 31, 2026What is cell design?Cell design refers to a method of assembling positive electrode material, negative electrode material, electrolyte, separator, and current collectors in certain proportions and pro...view
How are lithium battery cells designed? Which chemical system should be chosen?March 31, 2026What is cell design?Cell design refers to a method of assembling positive electrode material, negative electrode material, electrolyte, separator, and current collectors in certain proportions and pro...view Summary of Lithium Battery Export Data and Market Trends in the First Three QuartersMarch 31, 2026AbstractRecently, the European Union officially announced that it will impose a five-year anti-subsidy tariff on electric vehicles imported from China. Specifically, SAIC Group's products will be ...view
Summary of Lithium Battery Export Data and Market Trends in the First Three QuartersMarch 31, 2026AbstractRecently, the European Union officially announced that it will impose a five-year anti-subsidy tariff on electric vehicles imported from China. Specifically, SAIC Group's products will be ...view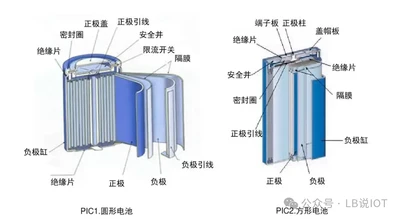 Safety First: A Multidimensional Strategy Analysis for Enhancing Lithium-ion Battery SafetyMarch 31, 2026Application and Safety Issues of Lithium-ion Batteries: Lithium-ion batteries are widely used due to their high energy density, high output power, and high average output voltage. However, accidents ...view
Safety First: A Multidimensional Strategy Analysis for Enhancing Lithium-ion Battery SafetyMarch 31, 2026Application and Safety Issues of Lithium-ion Batteries: Lithium-ion batteries are widely used due to their high energy density, high output power, and high average output voltage. However, accidents ...view Mechanism of SEI Film Formation in Lithium-Ion Batteries and the Impact of Formation Processes!March 31, 2026In the production process of lithium-ion batteries, formation is an extremely important step. This step mainly includes the initial charging process of the cell after the electrolyte injection is comp...view
Mechanism of SEI Film Formation in Lithium-Ion Batteries and the Impact of Formation Processes!March 31, 2026In the production process of lithium-ion batteries, formation is an extremely important step. This step mainly includes the initial charging process of the cell after the electrolyte injection is comp...view