Energy storage technology is fundamentally a technology that caters to demand, and its evaluation indicator system encompasses energy indicators, power indicators, scale indicators, lifespan indicators, efficiency indicators, self-discharge indicators, cost indicators, environmental impact indicators, and more. Depending on the application scenario, the types of indicators and their weights required by energy storage technology also vary.
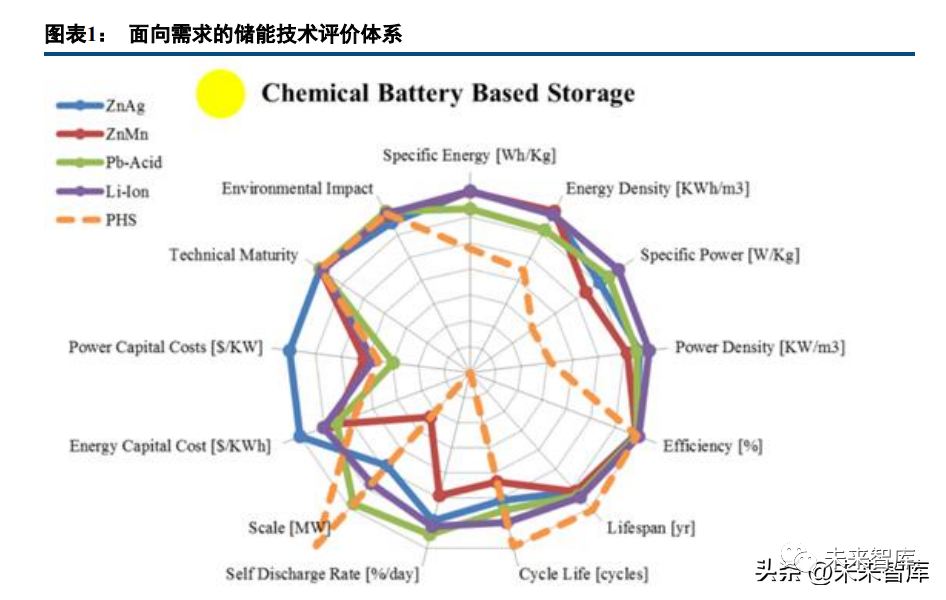
Taking consumer batteries, power batteries, and energy storage batteries as examples, consumer batteries tend to have higher volume and mass energy density, as well as higher charging rates; power batteries require a balance with high cost weight; energy storage batteries can relax energy and power-related indicators to some extent, but they have very high requirements for life and cost-related indicators. Among various energy storage technologies, secondary batteries (electrochemically rechargeable batteries) are a very critical component. They have a wide range of applications and a strong ability to interface with renewable energy power. From the perspective of applicable energy and power ranges, a variety of secondary batteries cover the vast majority of technical needs for energy storage applications, with lithium-ion batteries being the most universal. Overall, energy-related indicators corresponding to "storage capacity" are of the greatest importance at the level of secondary battery technology, and high specific energy (i.e., mass energy density) secondary batteries have the most potential applications, especially in cutting-edge scenarios; rate-related indicators corresponding to "throughput capacity" are also quite important. Of course, the performance of battery materials (sets) is the fundamental determinant of battery performance.
Although the basic concept is not complex, lithium batteries are actually a "multi-dimensional composite material" or "multi-functional system" that spans multiple spatial scales, involves various phase types, and continuously changes during use. We conduct classified research on different types of battery materials with a focus, and this focus must also consider the realizability of the overall performance of the battery.
3. Priority on energy density, with a balance and trade-off of rate performance and other aspects
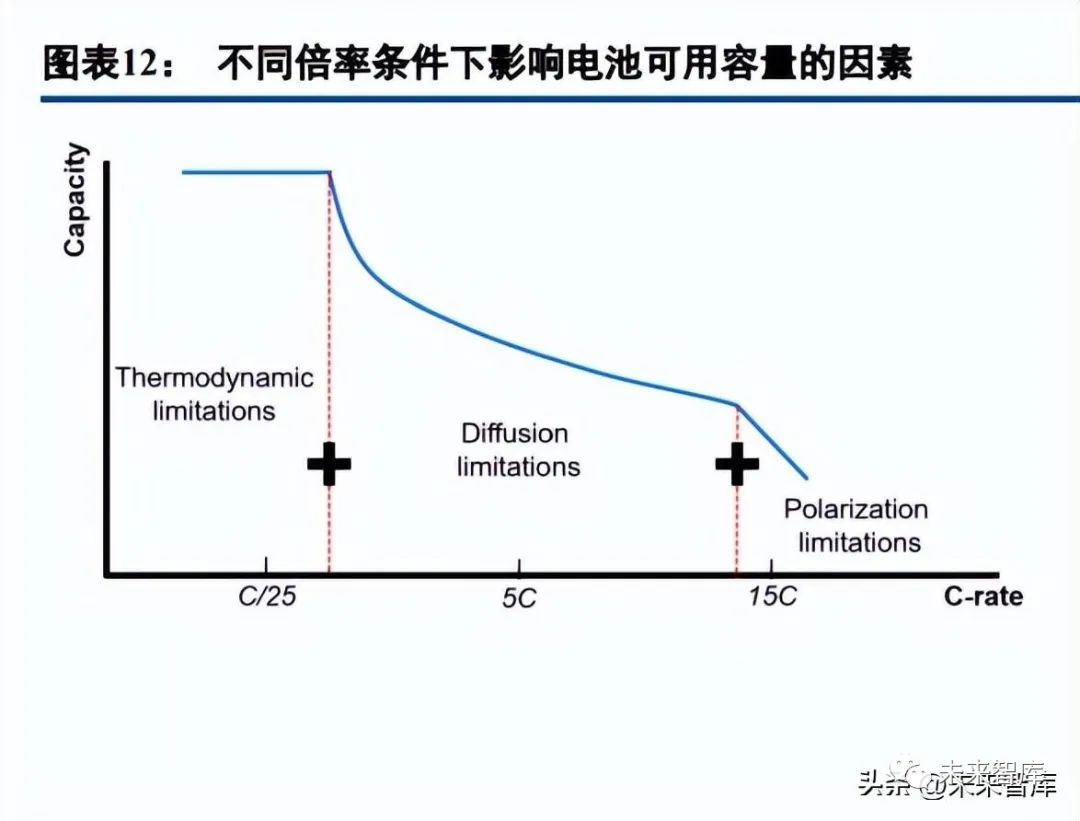
Lithium batteries are multi-dimensional composite systems spanning spatial scales and phase types, with material research needing to consider overall battery performance realizability. Improving lithium battery performance requires addressing degradation issues—high energy and rate capabilities amplify performance failure risks. High energy density relies on strong oxidizing cathodes and reducing anodes, which cause electrode volume changes and higher electrolyte demands; fast charging needs micro-macro optimization and material-system trade-offs. Temperature impacts performance significantly: low temperatures reduce efficiency, while high temperatures pose safety risks, making reasonable temperature control critical. Charged batteries have far lower safety than discharged ones, so various electrode and electrolyte modification methods are widely applied and continuously optimized.
Cathode vs. Anode, Symmetrical but Unequal
From the energy density formula E=U/[1/Qc+1/Qa+minact], cathode and anode specific capacities contribute symmetrically to energy density mathematically, but their material properties differ greatly. Phase-change materials have superior specific capacity but poor reaction kinetics and significant volume changes during lithium shuttling; intercalation materials are the opposite, and are the main type used in large-scale applications currently. Silicon-based phase-change anodes have limited practical use via doping, while phase-change cathodes (chlorides, sulfides, etc.) remain immature for application.
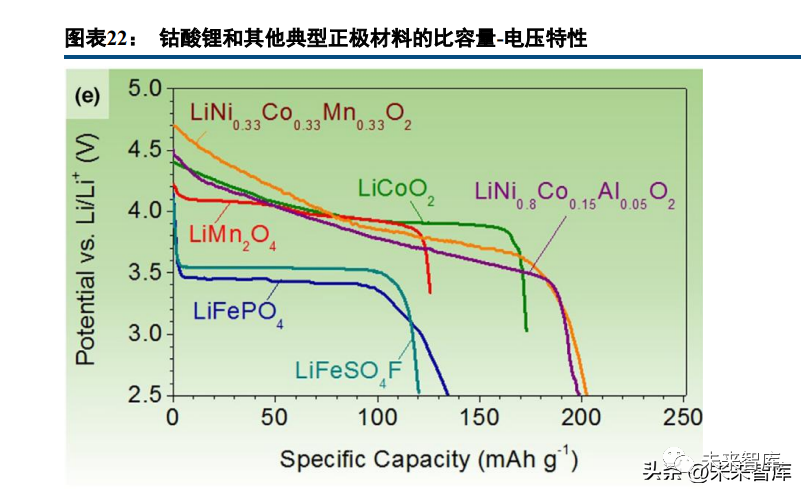
Graphite anodes offer a high specific capacity of nearly 370mAh/g (silicon-based anodes even higher), while high-voltage cathode systems (average >3V) have a specific capacity below 300mAh/g, making insufficient cathode specific capacity a key bottleneck for lithium battery performance. Taking lithium manganate as an example, the theoretical specific capacity calculation method for electrode materials includes five steps: defining lithiated/delithiated chemical formulas, calculating relative molecular mass, determining lithium substance change percentage, computing charge transfer via the Faraday constant, and dividing by molecular mass to get mAh/g. Lithium manganate has a theoretical capacity of 148mAh/g, lithium iron phosphate ~170mAh/g, and ternary lithium ~274mAh/g (actual capacity is related to theoretical capacity and lithium insertion/desertion boundary conditions).
Mainstream intercalation cathodes are divided into three systems by crystal structure—layered (lithium cobaltate, ternary materials), spinel (lithium manganate), and olivine (lithium iron phosphate)—all with oxygen ions as structural anions. Optimization of these materials is ongoing, and phase-change sulfur cathodes are also being developed to address the "capacity short board" of battery active materials.
Layered Oxide Cathode, Performance Optimization
Lithium cobaltate, the first commercialized layered oxide cathode, has a high theoretical specific capacity (274mAh/g), low self-discharge rate, and a cut-off voltage optimized to nearly 4.5V, remaining widely used in consumer electronics. However, cobalt is expensive, and lithium cobaltate has poor structural stability at high delithiation states, with thermal stability and high-rate cycle life as key concerns, driving continuous research into its modification.
Lithium nickelate has the same crystal structure as lithium cobaltate and uses cheaper nickel, but trivalent nickel is unstable (Jahn-Teller effect), and divalent nickel intermixes with lithium ions, causing irreversible phase transformations and poor thermal stability during cycling. NCA cathodes (doped with small amounts of cobalt and aluminum) improve electrochemical and thermal stability but may release oxygen under high pressure. Researchers have developed mixed transition metal ion layered cathodes, with the "nickel-manganese balance" plus cobalt doping (to suppress lithium-nickel mixing) being the most stable combination. The mainstream synthesis method is liquid-phase precursor preparation followed by calcination for lithium diffusion and valence control, leading to the birth of NCM ternary cathodes.
High-nickel NCM cathodes have a high proportion of tetravalent nickel at high delithiation states, achievable at low charging cut-off voltages—e.g., NCM811 has an available specific capacity >200mAh/g at 4.3V, and NCM9055 even higher. High-nickel cathodes boost battery energy density and have lower electrolyte pressure resistance requirements, but further increasing the cut-off voltage (e.g., to 4.8V) yields little effective capacity gain above 4.4V, and high delithiation causes irreversible phase transformations and cracking, reducing cycle life.
Medium-nickel cathodes (e.g., NCM424) show distinct characteristics: at 4.3V, the specific capacity is ~160mAh/g; at 4.5V, it exceeds 180mAh/g; and at 4.7V, it surpasses 220mAh/g. However, high voltage significantly impacts cycle life due to positive electrode irreversible phase transformation, oxygen release, and increased electrode-electrolyte interface side reactions.
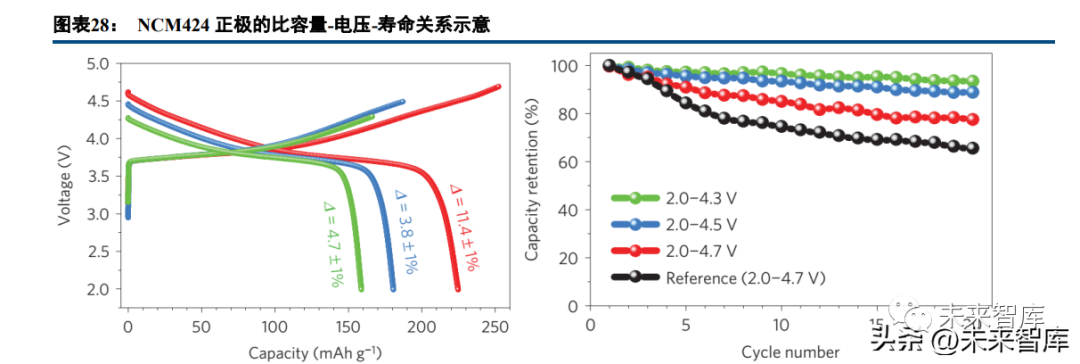
Comparing cathodes with different nickel contents, NCM9055 at 4.4V has a similar specific capacity to NCM111 at 5V, meaning high-nickel (standard voltage), medium-high nickel (higher voltage), and medium nickel (high voltage) cathodes yield similar battery energy densities. Medium-low nickel cathodes at ultra-high voltage are ideal for high energy density but require ultra-high electrolyte pressure resistance, with technical feasibility yet to be verified. Constructing a pressure-resistant cathode surface CEI film via electrolyte composition optimization and electrode coating/doping is an effective way to raise the actual charging cut-off voltage of ternary batteries.
Manganese aggravates lithium-nickel disorder (harming rate performance) but stabilizes lattice oxygen and layered structure (benefiting capacity retention), so ternary cathode transition metal ratios require comprehensive consideration. Ternary cathode particles can be polycrystalline (higher specific surface area, better rate performance) or single-crystalline. Bulk doping (e.g., aluminum for NCMA cathodes) reduces lattice deformation (-3.6% vs. >-4% for NCM/NCA), increases fracture strength (185.7MPa for NCMA vs. 125.5MPa for NCA and 137.2MPa for NCM), and improves cycle life by preventing cracking.

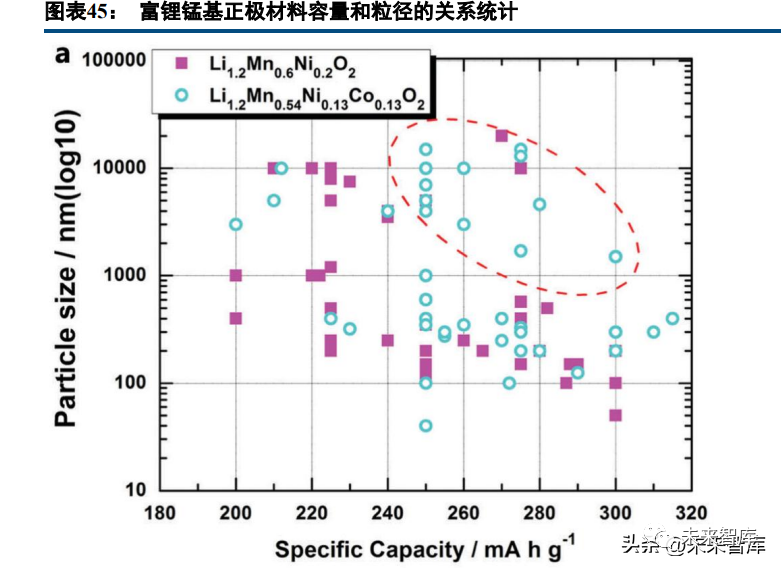
Lithium-rich manganese oxide (Li2MnO3) shows electrochemical activity above 4.5V; lithium-rich manganese-based oxides (xLiMO2·(1-x)Li[Li1/3Mn2/3]O2, formed by substituting nickel/cobalt for manganese-lithium pairs) have an initial reversible specific capacity >250mAh/g and a cut-off voltage close to 5V, attracting extensive research. Their key advantage is a specific capacity >250mAh/g at 2.0-4.8V, but capacity retention over long cycles is poor.
The first charge curve of lithium-rich manganese-based cathodes has two regions: below 4.5V, nickel/cobalt valence increases and lithium ions de-intercalate; above 4.5V, the Li2MnO3 layer contributes via manganese valence change, oxygen loss, and structural rearrangement. Post-first-cycle, oxygen migration causes lattice distortion, leading to voltage hysteresis, and continued cycling results in a continuous drop in average discharge voltage due to lithium/oxygen de-intercalation, transition metal dissolution, and irreversible spinel phase formation.
Lithium-rich manganese-based cathodes are similar to ternary cathodes in structure and composition, with synthesis methods including solid-phase, co-precipitation-calcination, and sol-gel-calcination (liquid-phase methods are preferred for complex structures). Composition strongly impacts performance: higher lithium content reduces average voltage, while higher cobalt content increases capacity. If technical issues are resolved, lithium-rich manganese-based cathodes are promising for high-energy density batteries at a 4.6V cut-off voltage. Key challenges include cycle life degradation from capacity/voltage changes, poor rate performance from lithium transport hindrance and thick CEI film formation. Modification methods (bulk doping, surface coating, treatment) aim to suppress oxygen release and side reactions and improve conductivity—common doping elements include K, Na, Co, Ti; coating agents include TiO2, Al2O3, and solid electrolytes; surface treatments include solution and atmosphere calcination.
Olivine Cathode, Advancing from Cost to Performance
Olivine-structured polyanion materials (with XO4³⁻ ion groups, X=P) have high lithium storage capacity and structural stability. Lithium iron phosphate (LFP), the classic olivine cathode, is widely used for its balanced performance and cost. LFP’s comprehensive performance can be optimized via particle morphology/size control and carbon coating (nano-sized particles and carbon nanotube doping improve rate performance but reduce compaction density).
Lithium iron manganese phosphate (LFMP) undergoes complete solid solution behavior and slight phase changes during lithiation/delithiation; phase change ranges impact capacity. Divalent manganese (0.083nm) and iron (0.078nm) have similar ionic radii, forming infinite solid solutions in the lithiated state, but trivalent manganese in the delithiated state distorts the metal-oxygen octahedron, changing lattice parameters and hindering lithium insertion/extraction.
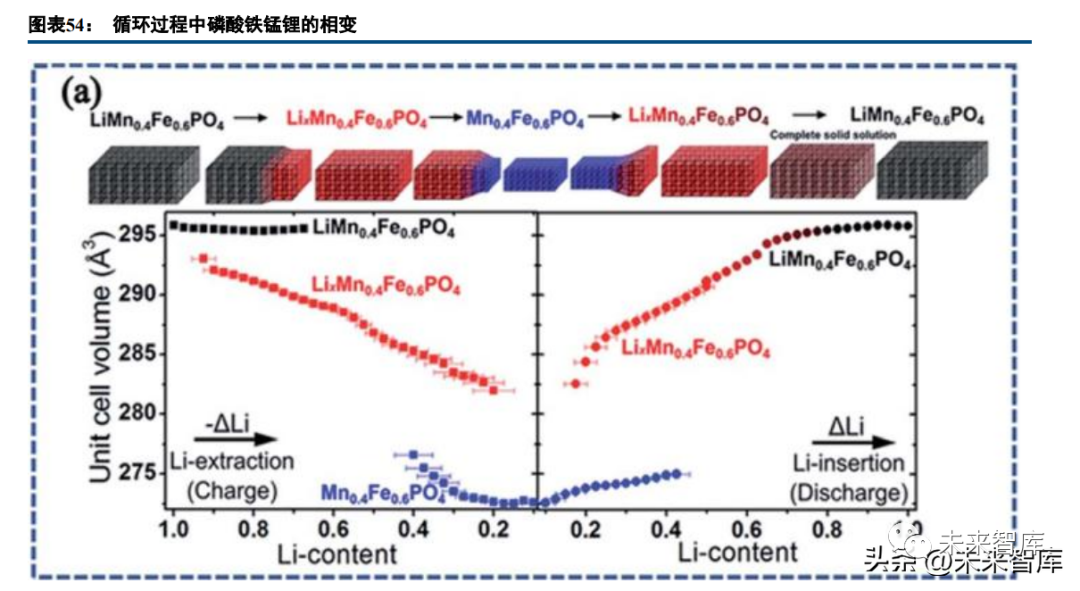
The iron-manganese ratio is critical for LFMP: high manganese content raises average voltage and energy density but damages the solid solution structure and reduces lifespan; a balanced ratio enables unobstructed lithium shuttling with a slightly lower voltage. Atomic-level uniform mixing of iron and manganese is essential—manganese micro-enrichment harms lifespan, rate performance, and conductivity.
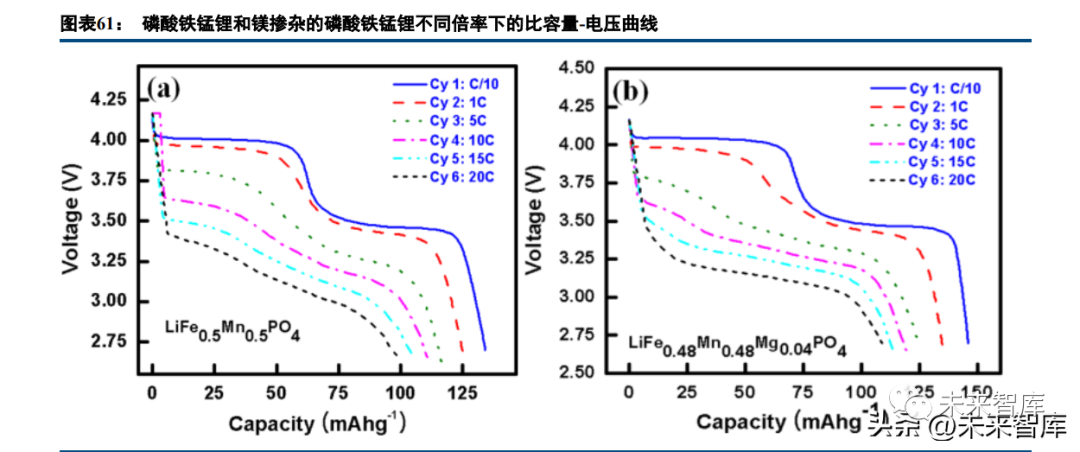
LFMP is synthesized via liquid-phase methods (sol-gel, co-precipitation) in laboratories and solid-phase methods in industry; sol-gel synthesis yields ~1μm particles with a carbon surface layer. Transition metal content impacts capacity: pure manganese cathodes have a high voltage platform but low effective capacity; adding 10% iron significantly improves capacity (single voltage platform); further iron addition creates two platforms (4V and 3.5V) with higher capacity. LFMP with a 1:1 iron-manganese ratio has a low-rate capacity >160mAh/g (two platforms), which degrades to ~130mAh/g at 1C (high voltage platform disappears).
Material performance is also affected by grain size, point defects, and modified element distribution. Carbon coating (especially carbon nanotube conductive networks) and magnesium doping (4% of transition metals, extending Li-O bonds to facilitate lithium migration) significantly improve conductivity and rate performance.
A high-performance LFMP cathode with an optimal iron-manganese ratio, uniform composition/particle size, reasonable doping, and a conductive coating can fully leverage its higher voltage advantage over LFP. Multiple listed companies have laid out LFMP technology, with medium/high manganese content products and ternary cathode doping/hybrid use expected to be the next breakthrough for olivine cathodes.
Spinel Cathode, Challenger Breaks Through the Wall
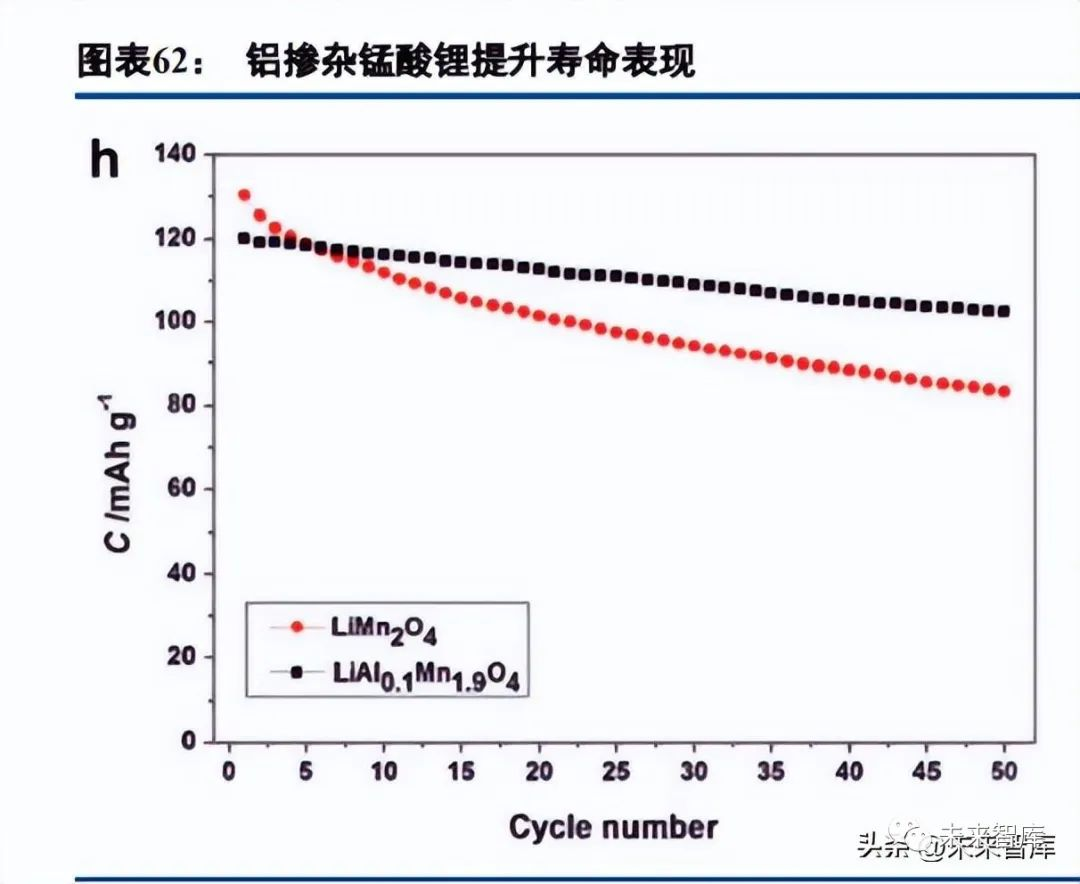
Spinel lithium manganate (LMO) is low-cost and has three-dimensional lithium diffusion channels with small volume changes, yielding good rate performance. However, +3 valence manganese causes J-T effect lattice distortion, particle cracking, and increased electrode-electrolyte side reactions; disproportionation and dissolution of +3 Mn lead to manganese deposition on the anode, accelerating electrolyte decomposition and SEI film thickening (consuming active lithium). Oxygen vacancies and high-temperature side reactions further degrade performance, resulting in shorter cycle life than LFP and ternary cathodes. LMO also has poor capacity/voltage performance: a theoretical specific capacity limit of 148mAh/g (lower than LFP’s 170mAh/g and layered cathodes’ 274mAh/g), an average voltage of 4V, and an average specific capacity of ~115mAh/g, leading to low system energy density. LMO is only suitable for cost-sensitive applications with low lifespan/energy density requirements (e.g., two-wheeled electric vehicles, low-speed EVs, A00-class models).
LMO modification methods include doping and coating (e.g., aluminum doping for better cycle life, TiO2 nanobelt doping for higher capacity). To significantly improve performance, a new material matrix is needed to boost voltage and optimize battery performance bottlenecks—high-voltage nickel manganese lithium oxide (LiNi0.5Mn1.5O4, LNMO) is the key breakthrough for spinel cathodes. Replacing 25% of Mn in LMO with Ni raises the voltage limit to 5V (platform ~4.7V), increasing battery energy density by ~20% (close to ternary batteries). LNMO has Mn mainly at +4 valence (less lattice distortion than LMO), but practical synthesis produces oxygen vacancies and +3 Mn (unavoidable lattice distortion); the high voltage also challenges conventional electrolytes (carbonate decomposition risk), and LNMO is susceptible to HF corrosion (causing Mn dissolution), requiring effective synthesis and modification.
LNMO is synthesized via the sol-gel method (high crystallization, good dispersibility but high cost, slow reaction) ; 300nm particles outperform 1μm particles in specific capacity and voltage at different rates (but uniform small particles reduce compaction density and increase side reactions). Bulk doping (Al, Cr, F, P, etc.) expands the solid solution region and improves structural/thermal stability; surface coating (carbon, oxides, organics) inhibits Mn dissolution and side reactions, boosting initial efficiency and cycle life. LNMO’s high voltage demands ultra-high electrolyte pressure resistance, a topic discussed in the electrolyte chapter.
Per unit energy cathode metal usage, LFP, LFMP, and LNMO save expensive transition metals and lithium; lithium-rich manganese-based cathodes have high lithium content but low expensive transition metal usage (based on Li1.2Ni0.16Co0.08Mn0.56O2).
Phase Change Material Sulfur Cathode, Taking an Unconventional Path
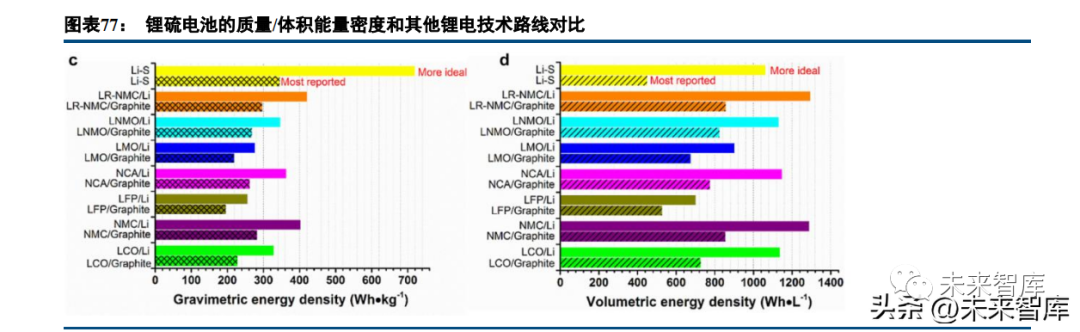
Sulfur cathodes are lithium-free (a key difference from other cathodes) and require a lithium-containing anode (typically lithium metal). Lithium-sulfur (Li-S) batteries have an extremely high theoretical specific energy density (2510Wh/kg) due to sulfur’s high capacity (1672mAh/g). However, sulfur has low lithium-ion diffusion/electronic conductivity and significant volume changes during lithium storage/release, which can be mitigated by composite material systems (e.g., carbon-sulfur composites) at the cost of partial specific capacity.
During Li-S battery discharge, Li2S forms via Li2Sx intermediate products; these polysulfides "shuttle" to the anode, oxidizing lithium metal, and reduced polysulfides shuttle back to the cathode, causing capacity loss, low coulombic efficiency, and high self-discharge. Suppression methods include adding lithium nitrate, regulating electrode composition/morphology, and constructing physical barrier layers; solid electrolytes fully suppress the shuttle effect and improve safety—e.g., a PEO-LLZO composite solid electrolyte maintains a cathode capacity >800mAh/g after 200 cycles (comparable to liquid systems).
Current Li-S battery research reports a maximum mass energy density of 300Wh/kg (theoretical density far higher than mainstream systems), but volumetric energy density is poor (even lower than LFP-graphite batteries).
Graphite Anode, No Doubt about Specific Capacity, Rate Capability as a Plus
Lithium graphite intercalation compounds were synthesized in the 1950s; with suitable electrolytes, a stable SEI film forms on the graphite surface, enabling reversible lithium intercalation/deintercalation. Graphite has a balanced performance with a specific capacity of 372mAh/g, making artificial/natural graphite the mainstream lithium storage anode (special graphites like graphene have higher capacities). The low lithium storage potential of graphite and stable SEI film formation ensure system stability; lithium plating is related to temperature (hot spots at low temperatures trigger plating).
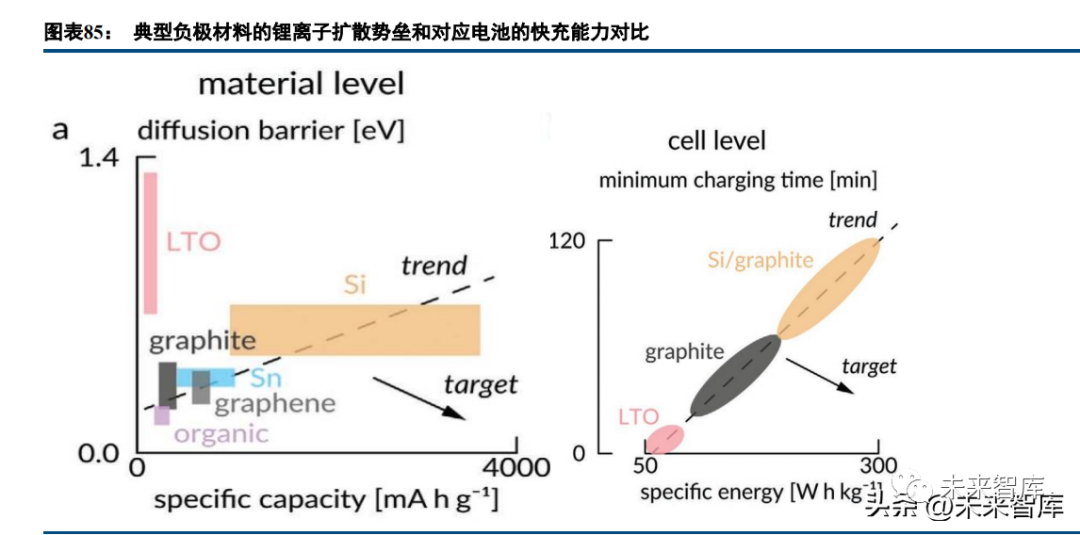
With practical capacity close to the theoretical limit, graphite anode research now focuses on rate performance (especially fast charging). Graphite is suitable for EV fast charging from a diffusion barrier perspective, but its layered structure (lithium intercalation only from end faces) and small specific surface area limit fast charging capability (a large specific surface area reduces initial efficiency). Researchers optimize fast charging via constructing graphite/composite anodes with appropriate orientation, particle size, porosity, surface modification (oxidation, amorphous carbon coating), and doping (P, B), balancing specific capacity, initial efficiency, and fast charging. Electrolyte adaptation and conductive agent selection are also critical.
Silicon-based Anode, Leveraging High Specific Capacity to Address Shortcomings
Silicon has an ultra-high theoretical specific capacity: 4200mAh/g (Li22Si5, high temperature) and 3579mAh/g (Li15Si4, room temperature), with a volumetric capacity of 9786mAh/cm3 (vs. 837mAh/cm3 for graphite) and a low delithiation voltage (~0.5V, slightly higher than graphite). Silicon-based anodes are ideal for high-energy density lithium batteries, especially when paired with high-nickel NCM/NCA cathodes.
However, silicon undergoes massive volume changes during lithiation, causing particle cracking, loss of contact with conductive agents, and poor cycle life (critical particle size ~150nm). Silicon also forms a loose, thick, high-impedance SEI film (unlike the dense SEI of graphite), leading to low initial efficiency and repeated SEI peeling/regeneration during cycling (consuming active silicon/lithium and degrading performance).
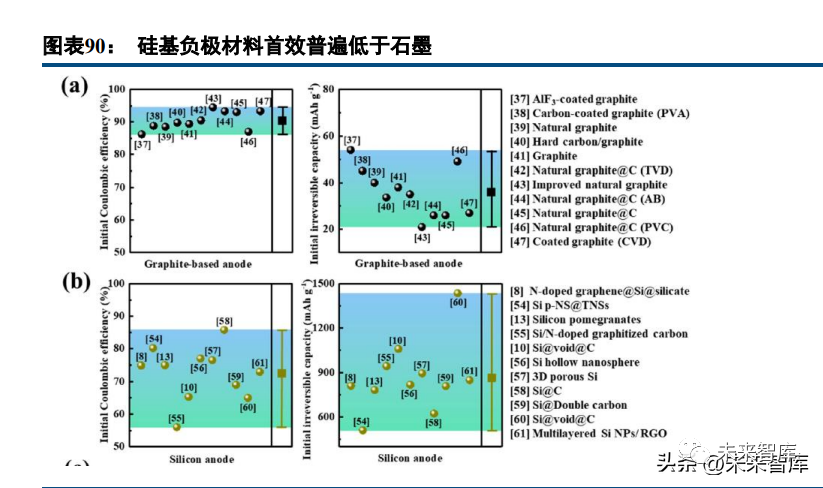
Silicon-based anode technical routes include silicon-carbon, silicon oxide-carbon, and silicon alloys, with silicon-carbon anodes being the most practical. Lithiated silicon-carbon composites show lithium enrichment in surface nanopores and a Li-Si-C mixture in the interior; lithium migrates in the order of C → nanopores → Si during lithiation (alloying with Si causes significant volume changes). For silicon oxide anodes, elemental silicon is the active lithium storage component; Li2O acts as a fast ion channel, and lithium silicates (Li2O, Li4SiO4) buffer volume changes (benefiting rate performance and cycle life). However, these silicates are inert phases, leading to lower theoretical capacity and first-cycle efficiency (e.g., silicon monoxide: 1480mAh/g, 70.9% first efficiency) than silicon-carbon anodes, and even lower than graphite, ultimately reducing battery energy density.
To improve silicon-based anode specific capacity, rate performance, and cycle life for high-performance power batteries, optimization is needed for silicon/silicon monoxide synthesis/modification (particle size control, carbon composite/coating, pre-lithiation), as well as electrolyte, conductive agent, and binder adaptation. Silicon-based anodes are high-potential but challenging, with ongoing scientific, engineering, and commercial efforts.
Lithium Metal Anode, the Holy Grail of Thorns
Lithium metal has a theoretical specific capacity of 3860mAh/g and a 0V voltage (infinite capacity if lithium is from the cathode), earning it the title of the "holy grail" of anodes. Research on lithium metal anodes predates commercial lithium-ion batteries: M. Stanley Whittingham invented the Li-TiS2 battery in 1970, and Moli Energy mass-produced lithium metal anode secondary batteries in 1985 (100Wh/kg, used in 3C products). However, safety incidents (fire/explosion) led to NEC’s acquisition of Moli Energy in 1990.
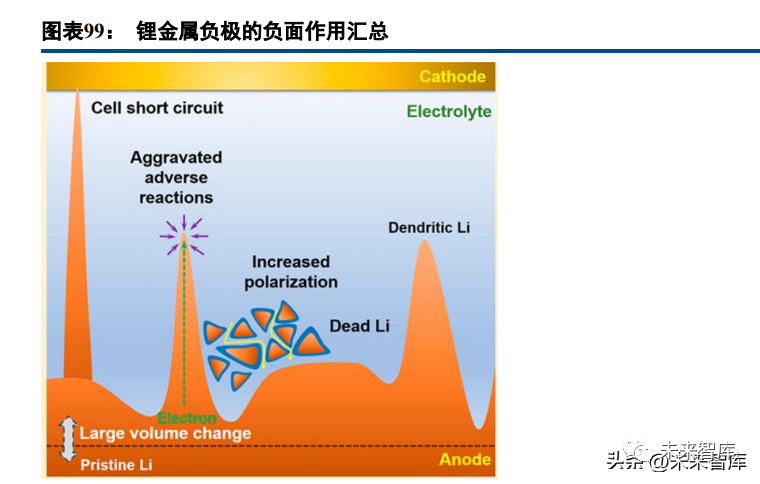
NEC found that uneven lithium deposition (lithium dendrite formation) and dendrite piercing of separators cause capacity decay, short circuits, and safety hazards. Side reactions between lithium and electrolytes, as well as lithium volume/morphology changes, also impact performance. While graphite anodes are industrialized, lithium metal anodes remain a research focus: phenomenon/mechanism observation, electrolyte system matching, electrode modification, and "anode-free batteries" (lithium from the cathode, only present in the charged state at the anode).
Lithium metal forms an unstable SEI film with conventional electrolytes, exacerbating dendrite growth, side reactions, and volume changes. Key research points include lithium usage amount, electrolyte system design, and lithium passivation. Battery cycling shows linear capacity decay from lithium-electrolyte side reactions and sudden capacity drops from poor SEI layer growth; excess lithium consumes electrolyte (forming "dry SEI"), while anode-free batteries have dead lithium in the SEI (degrading performance). A 20μm lithium foil forms a stable "wet SEI" film, yielding excellent experimental results: 600 effective cycles and 350Wh/kg.
Electrolytes are critical for lithium metal anodes—high-performance systems use LiFSI as the lithium salt and fluorinated/ether/sulfonamide solvents. Solid electrolytes (hard, dense, stable) can suppress dendrite growth, but polymers may be pierced by dendrites, and inorganic solid electrolytes have dendrite deposition in gaps.
Electrolyte, Conducting Lithium to the Electrodes
Electrolytes are the "lithium-ion transport river" for lithium batteries, requiring high ion conductivity, electronic insulation, good electrode contact, oxidation/reduction resistance, electrochemical/thermal/air/mechanical stability, wide temperature adaptability, and low cost. Due to the high inter-electrode potential difference (exceeding water’s decomposition voltage), mainstream electrolytes are organic solvent-lithium salt systems with an ion conductivity of ~10⁻³~10⁻² S/cm (excluding expensive ionic liquids).
Electrolyte solvents dissolve lithium salts and require high polarity (dielectric constant), a wide electrochemical window, low viscosity, and heat resistance. No single solvent meets all requirements, so mixed solvent systems are used: high dielectric constant solvents (EC, PC) paired with low viscosity solvents (DMC, DEC, EMC). Solvent additives (VC, FEC, DTD, etc.) enhance lithium ion solvation, stabilize SEI films, and assist flame retardancy. Researchers are developing fluorinated carbonate solvents (higher oxidation potential) and non-carbonate systems for lithium metal anodes and high-voltage cathodes.
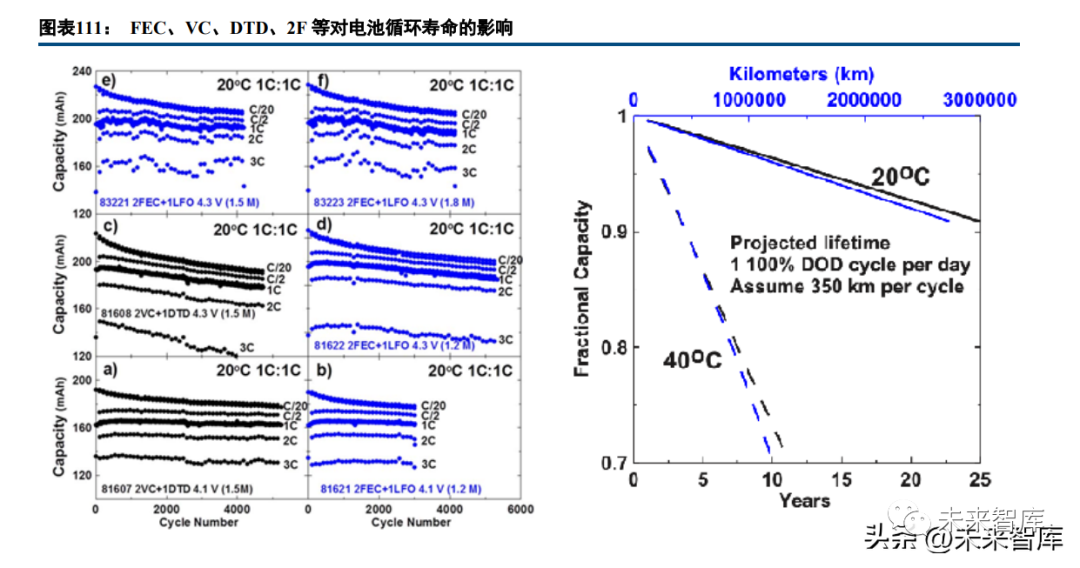
Innovative electrolyte designs include single fluorinated solvent FDMB (paired with LiFSI) for NCM-lithium metal batteries (99.98% coulombic efficiency under excess lithium, 325Wh/kg initial energy density for NCM811 anode-free batteries) and sulfonyl solvent DMTMSA (paired with LiFSI) with fewer side reactions, less gas production, and stable lithium metal morphology (only ~10% capacity decay after 100 cycles at 3.0-4.7V).
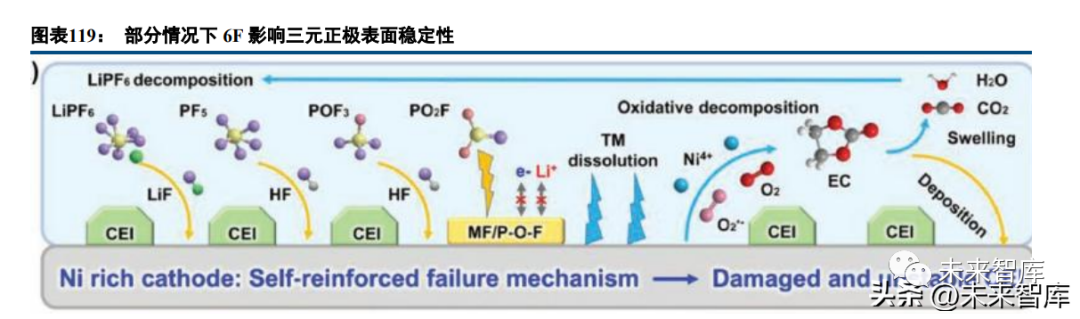
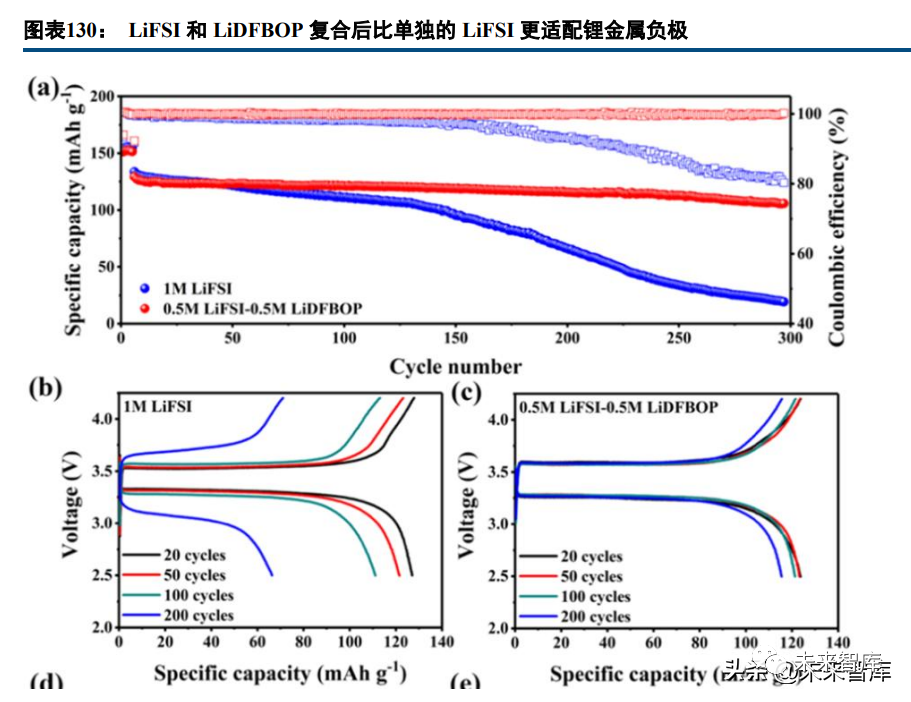
Lithium salt additives (LiDFP, LiBOB, LiDFOB, etc.) optimize the electrolyte and electrode-electrolyte interface. LiPF6 decomposition produces HF (harming battery performance), so new lithium salts are being researched. LiBOB passivates high-voltage cathode surfaces, consumes HF, and forms a stable CEI film; dual salt systems (e.g., LiFSI-LiDFBOP) perform better for lithium metal batteries than single salts.
Ionic liquids are an important high-performance electrolyte branch with good thermal stability, non-volatility, a wide electrochemical window (up to 5V), non-flammability, and high ion conductivity.
Solid Electrolyte, Looking Forward Together, the Eternal Spring
Solid-state batteries are broadly classified by liquid phase content: quasi-solid, semi-solid, solid, and all-solid (no liquid phase). Solid electrolytes have the same performance requirements as liquid electrolytes (ion conductivity, stability, etc.) and are divided into polymer and inorganic types.
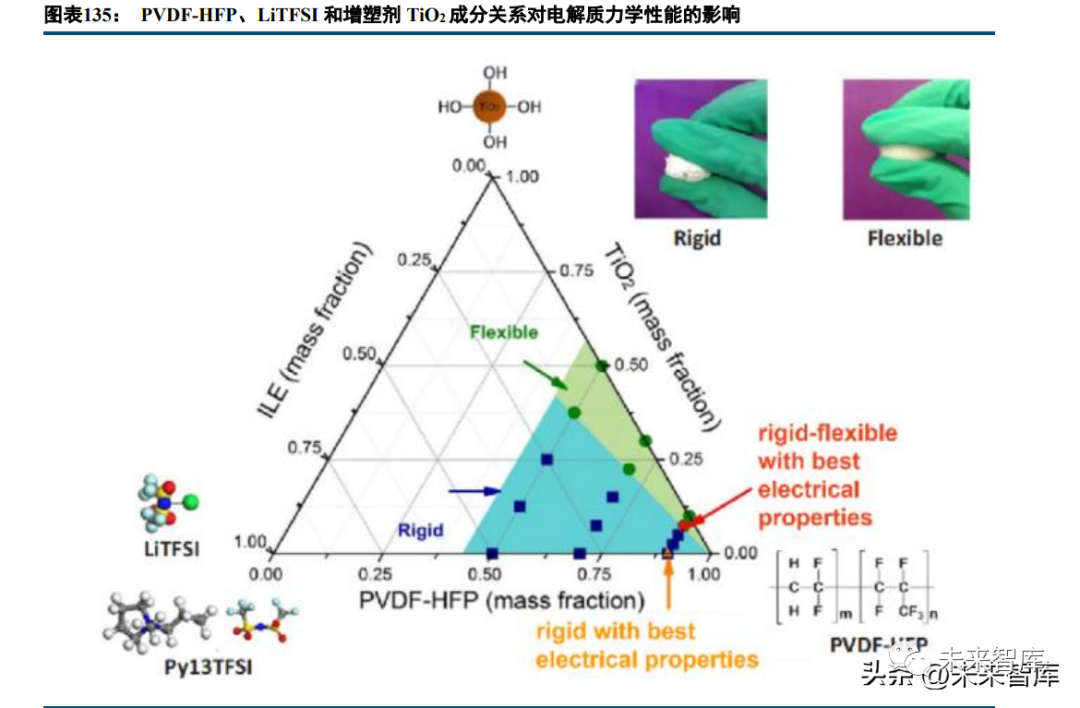
Polymer solid electrolytes: Lithium ions migrate in the amorphous region via polymer segment movement and complexation/decomplexation. Optimization focuses on reducing the glass transition temperature and expanding the amorphous region (plasticization). Main matrices include PEO (most studied, high dielectric constant, good electrode contact but low room temperature ion conductivity ~10⁻⁸~10⁻⁶ S/cm; modified PEO reaches 10⁻⁴ S/cm), polysiloxane (good thermal stability, low Tg but poor lithium salt solubility), polycarbonate (high dielectric constant, 10⁻⁵~10⁻⁴ S/cm but average oxidation resistance), and PVDF (high dielectric constant, chemical stability but average conductivity). Lithium salt doping (sulfonylimide salts for high conductivity) and inorganic filler addition (SiO2, TiO2; reduce crystallinity, form lithium transport channels) improve performance. Single ion polymers and polyelectrolyte liquids are forward-looking directions.
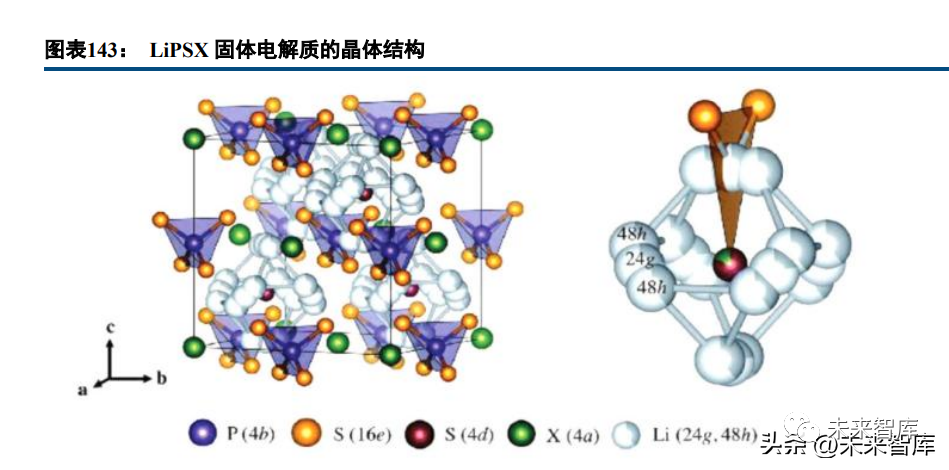
Inorganic solid electrolytes: Lithium ions migrate between anion-framework lattice structures, classified by main anions into oxides, sulfides, halides, etc.
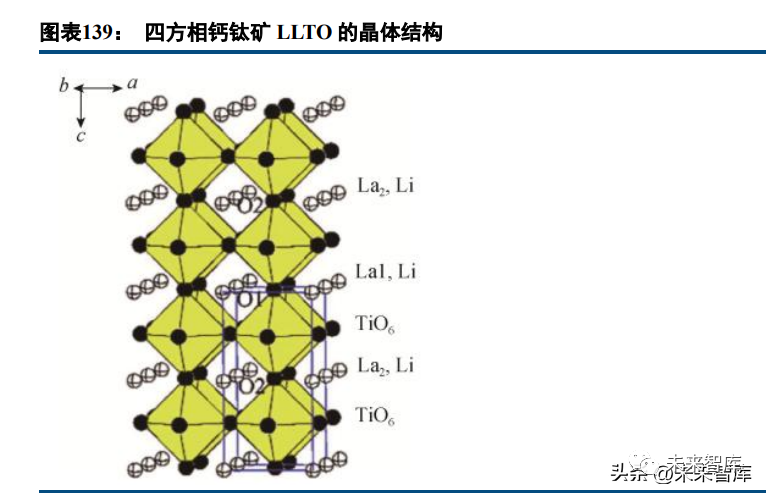
• Oxides: LLTO (perovskite, 10⁻⁴~10⁻³ S/cm but unstable to lithium metal), NASICON (10⁻⁴~10⁻³ S/cm, LAGP more stable than LATP), LLZO (garnet, 10⁻⁴~10⁻³ S/cm, good chemical stability), and LiPON (amorphous film, 10⁻⁶ S/cm, suitable for thin-film batteries).
• Sulfides: Larger lithium diffusion channels and weaker electrostatic binding than oxides yield higher conductivity (Li10GeP2S12 ~10⁻² S/cm). LGPS has high conductivity but poor stability; Li7P3S11 and Li6PS5X (X=Cl/Br) have improved conductivity (10⁻²~10⁻³ S/cm). Sulfides are softer than oxides but sensitive to water/oxygen and form space charge layers with layered oxide cathodes.
• Halides: Potential for high conductivity due to large diffusion channels and weak lithium binding; strong oxidation resistance but sensitive to humidity and lithium metal.
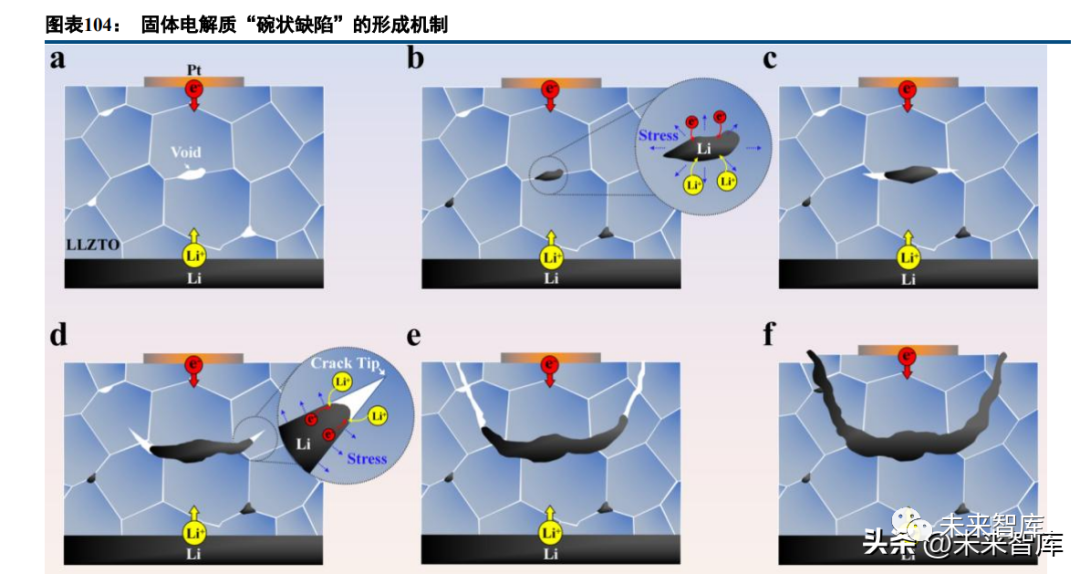
Solid electrolytes have advantages (high thermal stability, wide electrochemical window) but also weaknesses: no single type achieves satisfactory comprehensive performance, with low room temperature ion conductivity and poor electrode contact as key issues (sulfides have conductivity comparable to liquid electrolytes, but interface resistance remains high). Modification strategies include electrode surface treatment (lithium metal, ternary cathodes), solid electrolyte composites, and solid-liquid electrolyte hybrids (gel electrolytes with 10⁻³ S/cm room temperature conductivity, higher volumetric energy density).
A "polymer-ceramic-polymer" composite solid electrolyte (NASICON coating + PE separator + LiTFSI-doped PEO) shows excellent performance: LFP-lithium metal batteries have almost no capacity decay after 200 cycles at 0.2C (60°C) and high safety after folding/shearing, due to strong membrane structure and mixed conduction interfaces (suppressing dendrites).
Solid-state battery production processes overlap with liquid batteries in front-end steps but require additional steps for lithium metal anodes and solid electrolyte coating, and pressure/sintering in middle/back-end steps (no liquidation). Industry strategies focus on leveraging safety advantages, improving energy density, optimizing rate/cycle life/processability, and targeting high-safety special applications and high-end EVs. With performance and cost progress, solid-state batteries may become a key lithium battery technical route.
Pre-lithiation
Irreversible lithium loss during battery formation and cycling degrades performance; pre-lithiation compensates for this loss, optimizes the N/P ratio, and enhances actual energy density and lifespan by utilizing active lithium fully. Pre-lithiation mainly addresses anode SEI film lithium consumption, with anode lithium supplementation (direct replenishment) being the most common method.
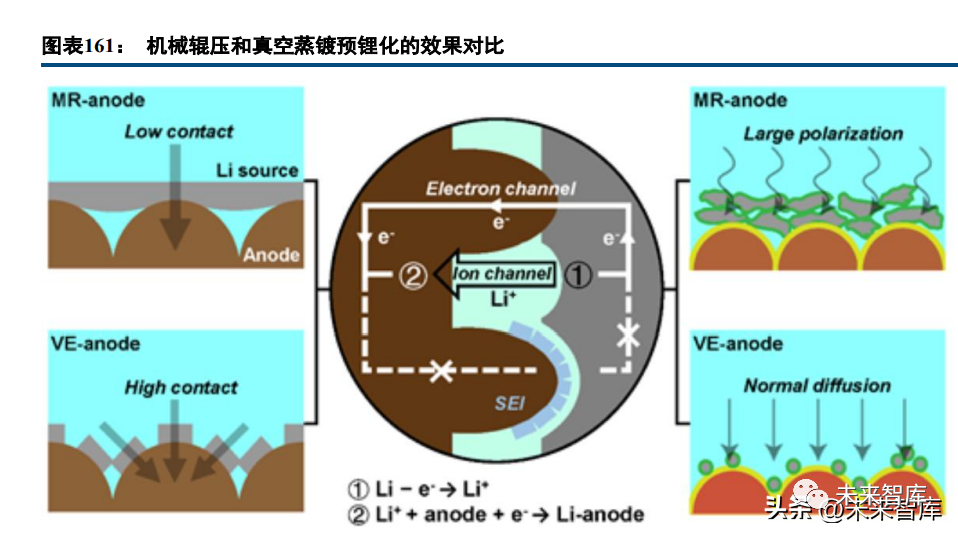
Anode lithium supplementation methods include: adding surface-stabilized lithium powder (high capacity ~3860mAh/g, small dosage but high chemical reactivity, safety risks, and strict production/transport standards); lithium foil contact (difficult pre-lithiation control, high foil technical requirements); and lithium alloys (Li-IV group metals, low voltage, high capacity, better stability than pure lithium but similar reactivity issues). Unutilized lithium sources form dead lithium, hindering lithium diffusion and causing plating; enhancing interface "electronic pathways" (e.g., vacuum thermal evaporation instead of mechanical roller pre-lithiation) improves pre-lithiation efficiency.
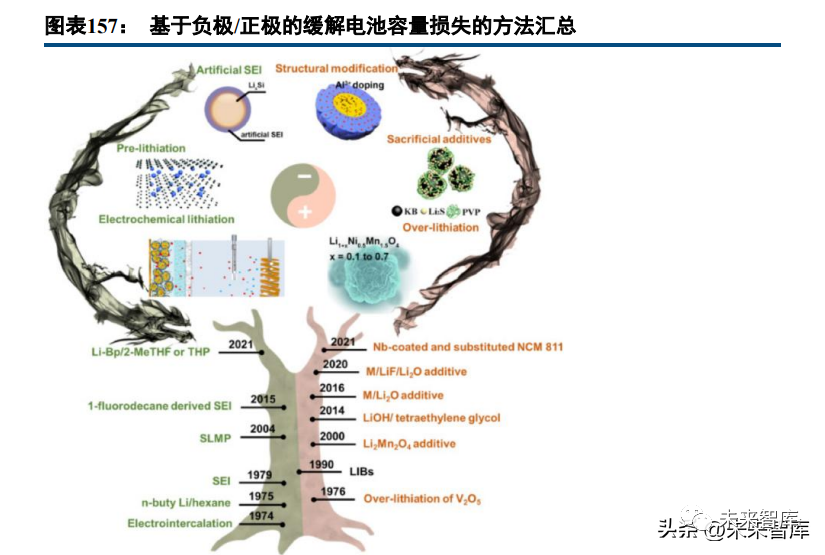
Other pre-lithiation methods include chemical pre-lithiation (reducing agent treatment to transport lithium to the anode) and electrochemical pre-lithiation (external voltage-driven lithium ion diffusion from lithium foil to the anode). Cathode lithium supplementation (indirect replenishment) pushes excess lithium from cathode replenishers/lithium-rich cathodes to the anode during charging, with higher compatibility to existing manufacturing processes (direct doping of replenishers or lithium-rich cathodes). Cathode replenishers include lithium oxides/nitrides/sulfides (lithium-rich composite oxides like Li2NiO2 and Li5FeO4 are preferred for balanced capacity and processability).
Cathode replenishers have higher chemical stability than anode replenishers but face issues like slurry gelation, gas production during pre-lithiation, and residual low-capacity materials after lithium release (impacting battery performance). Cathode supplementation is suitable for high-safety, process-compatible scenarios with low lithium demand; anode supplementation is better for large-capacity replenishment. Lithium-rich metal salt-based cathode replenishers are more mature, while metallic lithium-based anode replenishers are in competitive development. Pre-lithiation technology can significantly enhance the competitiveness of high-energy density/long-life batteries.
Conductive Additives, the Stage for Carbon Materials
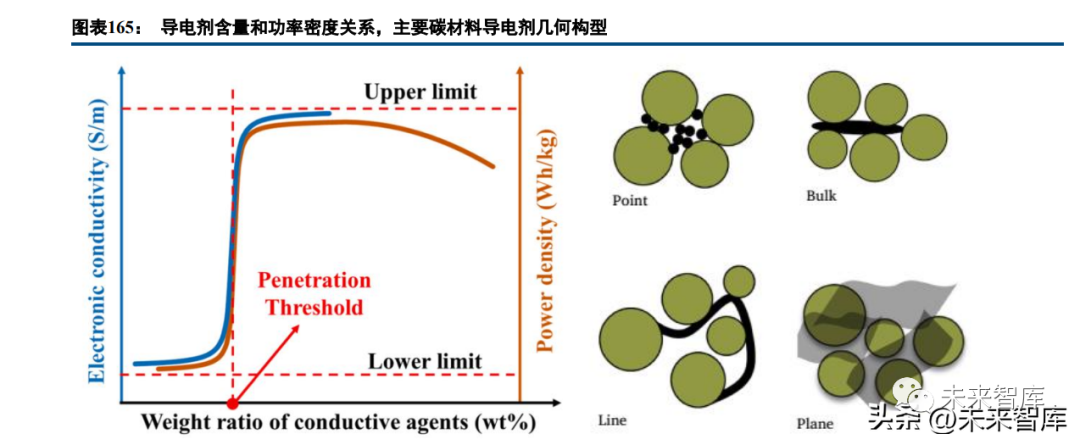
Effective electron transmission requires high bulk/interface electronic conductivity; conductive additives in electrodes are critical for optimizing this performance. Carbon nanotubes (CNTs) significantly improve battery material electronic conductivity compared to conventional carbon black, and CNT-high-end carbon black composites yield better results. Higher aspect ratio CNTs boost conductivity, but dosage/proportion must balance performance and cost. CNTs remain the representative of high-end conductive additives and will gradually penetrate mid-low-end markets with cost control.
Sodium-ion Battery, The Eve of Blossoming Just Below the Top
Lithium resource supply gaps and price surges, combined with growing demand for consumption/power/energy storage, have accelerated sodium-ion battery (SIB) research. Sodium and lithium ions have similar properties, so SIB electrolyte development follows lithium-ion battery (LIB) routes: aqueous and organic electrolyte systems have better bulk ion conductivity than solid electrolytes (opposite to LIBs). Aqueous electrolytes have high conductivity but a narrow electrochemical window (1.23V vs. >4V for carbonates), reducing energy density; organic electrolytes (carbonate solvents + sodium salts) are the most suitable for SIBs.
Graphite, the mainstream LIB anode, cannot store sodium ions (it stores lithium and potassium), which was a key reason for SIBs failing to industrialize with LIBs in the last century.
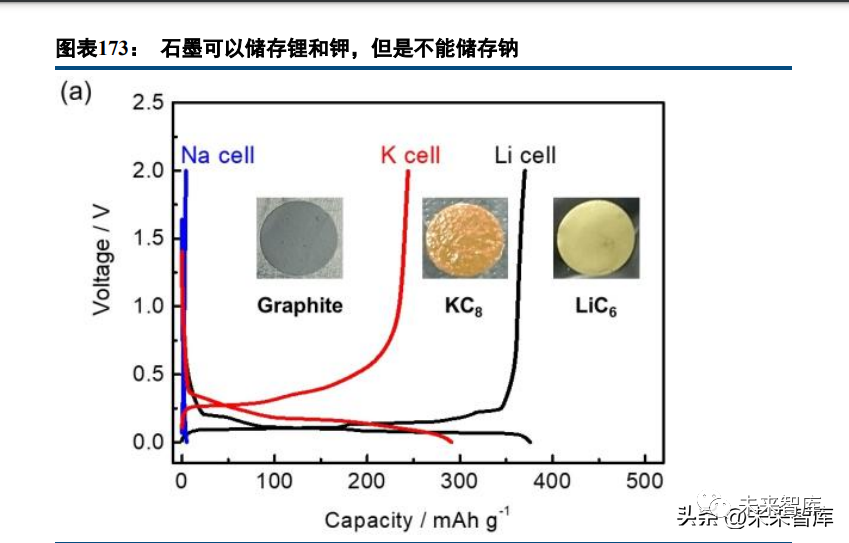
SIB cathode materials have a practical capacity of tens to >200mAh/g and a voltage of 2~>4V; sodium ion diffusion and material stability (composition, valence, phase) are critical for performance. The most reasonable short-to-medium term SIB material system is: hard carbon anode (dominant), layered oxide/Prussian blue/polyanion cathode, organic carbonate-sodium salt electrolyte, separator, and aluminum foil current collector. SIB production processes/equipment are identical to LIBs (unlike solid-state/Li-S/lithium-air batteries), reducing industrialization costs.
SIBs have advantages over LIBs in low-temperature performance (simple desolvation) and safety (aluminum foil dual current collector). Conventional SIBs compete with LFP via cost advantages and comparable performance; high-energy SIBs may exceed LFP in energy density but have low technology maturity and high uncertainty (volumetric energy density scaling is more difficult). SIBs outperform lead-acid batteries in all performance indicators except extreme safety, with a narrowing cost gap after full development. SIBs are a promising secondary battery, with applications in two-wheeled EVs, low-speed EVs, energy storage, and even power batteries.
High-valence Carriers, a Long and Arduous Journey
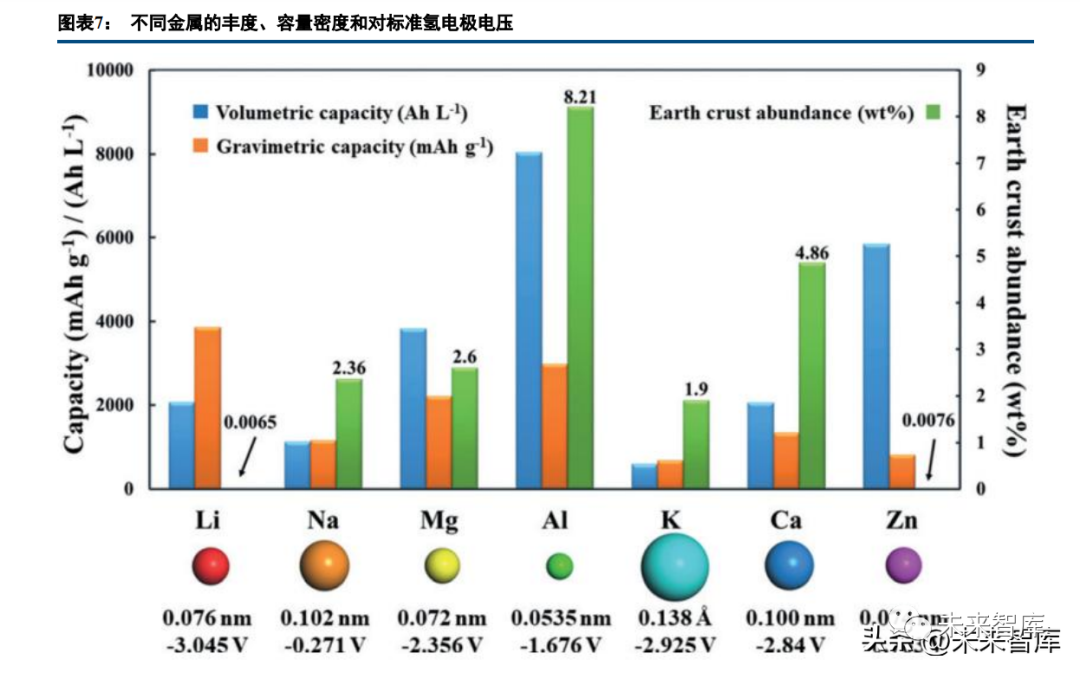
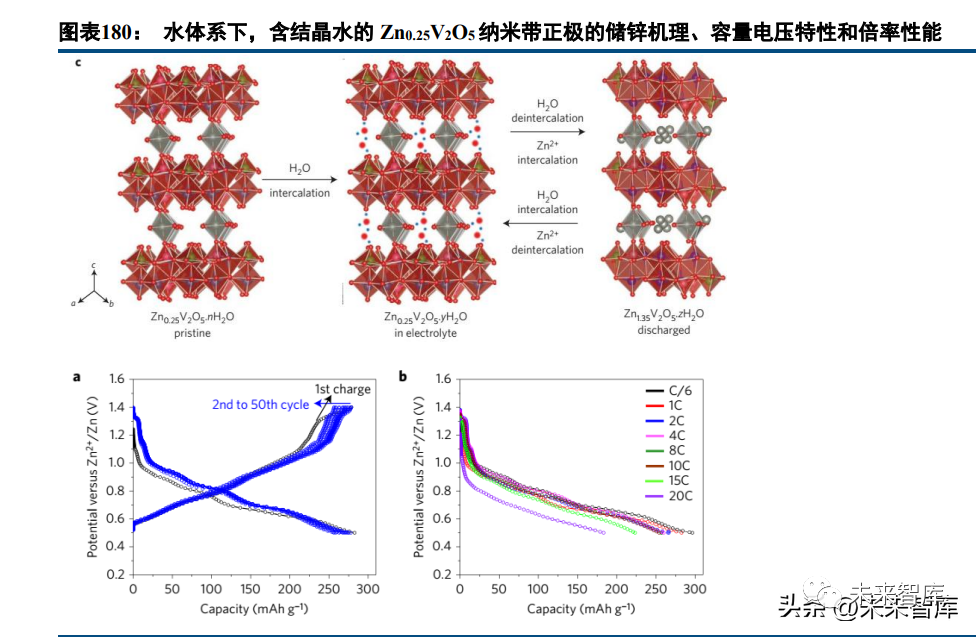
Research on batteries with +2/+3 valence carriers (Mg, Al, Zn) is ongoing, but cathode/electrolyte development is highly challenging, with anode modification also required, making practical application far off. Magnesium-ion batteries with chelated magnesium ions in organic systems have a theoretical energy density of 412Wh/kg; vanadium-based oxide cathodes for aqueous zinc-ion batteries show a high specific capacity (300mAh/g), good rate performance, and long cycle life (~1000 cycles).
All-vanadium redox flow batteries (hydrogen ion carriers) have excellent cycle life (>10,000 cycles) and safety but suffer from low energy density, low energy cycle efficiency, and high initial costs.
Nobel Prize, The "First Victory" in the Energy Revolution
Lithium-ion battery development spans nearly a century, from concept proposal to material system completion, with numerous disruptive research results and industrial efforts (including safety accidents from early lithium metal battery applications). Technology, policy, and market drive each other in lithium battery development: consumer batteries first replaced Ni-Cd/Ni-MH batteries, enabling portable electronics; power batteries opened the EV era, challenging the traditional fuel car industry; energy storage batteries are now competing with pumped storage technology.
In 2019, the Nobel Prize in Chemistry was awarded to John B. Goodenough, M. Stanley Whittingham, and Akira Yoshino for their pioneering contributions to lithium battery research and development. The Nobel Committee noted that lithium batteries are widely used in mobile phones, laptops, EVs, and renewable energy storage, laying the foundation for a fossil fuel-free society.

Forward with Courage, a Shining Future
Lithium battery technology still has huge room for progress:
• Short-to-medium term: Super high-nickel/higher voltage ternary cathodes, LFMP cathodes, high-silicon anodes, pre-lithiation, and optimized electrolytes/conductive agents will drive LIBs toward "specialized and multi-functional" performance.
• Medium-to-long term: LNMO cathodes, lithium-rich manganese-based cathodes, lithium metal anodes, high-performance electrolytes/solid electrolytes are key research directions; SIBs will become an important cost-effective niche route; other high-valence carrier batteries have breakthrough potential.
"Next-generation batteries" have a bright future, and lithium-ion batteries (and new battery types) will eventually reach their performance-cost ceiling, as with all great technological achievements. High-performance, low-cost batteries will become the cornerstone of the energy revolution, driving human civilization progress—this "taken for granted" progress is the result of continuous creation, and will be remembered in the history of civilization.
 Celebrating 2 Years: JREPower Indonesia Production Base AnniversaryJanuary 13, 2026Today, JREPower (Shenzhen JREPower Tech Co., Ltd.) grandly celebrated the 2nd anniversary of its production base establishment in Jakarta, Indonesia. The milestone event gathered local government...view
Celebrating 2 Years: JREPower Indonesia Production Base AnniversaryJanuary 13, 2026Today, JREPower (Shenzhen JREPower Tech Co., Ltd.) grandly celebrated the 2nd anniversary of its production base establishment in Jakarta, Indonesia. The milestone event gathered local government...view A Guide to Lithium Battery Energy StorageMarch 31, 2026Why are lithium batteries so widely used, what are their advantages, and what are the main applications at present? The following content will sort out the concept definition, working principle, indu...view
A Guide to Lithium Battery Energy StorageMarch 31, 2026Why are lithium batteries so widely used, what are their advantages, and what are the main applications at present? The following content will sort out the concept definition, working principle, indu...view Analysis and Demand Forecast of Consumer Lithium Battery Market in 2024!March 31, 2026Part 1: Steady Growth in Traditional Consumer Electronics1.1 Smartphones: The global shipment growth rate is expected to be 4% in 2024. According to IDC, the global smartphone shipment growth rate is ...view
Analysis and Demand Forecast of Consumer Lithium Battery Market in 2024!March 31, 2026Part 1: Steady Growth in Traditional Consumer Electronics1.1 Smartphones: The global shipment growth rate is expected to be 4% in 2024. According to IDC, the global smartphone shipment growth rate is ...view Introduction to Lithium-ion Battery Standards (International Edition)March 31, 2026I. UN38.3UN38.3 is the section of Chapter 38, Paragraph 3 of the United Nations' "Manual of Tests and Criteria for the Transport of Dangerous Goods." In 2003, the United Nations adopted ...view
Introduction to Lithium-ion Battery Standards (International Edition)March 31, 2026I. UN38.3UN38.3 is the section of Chapter 38, Paragraph 3 of the United Nations' "Manual of Tests and Criteria for the Transport of Dangerous Goods." In 2003, the United Nations adopted ...view Understanding the Impact of High and Low Temperatures on Lithium Battery PerformanceMarch 31, 2026Lithium iron phosphate batteries and ternary lithium batteries in lithium batteries have the advantages of high energy density, wide operating temperature range, long cycle life, and safety and reliab...view
Understanding the Impact of High and Low Temperatures on Lithium Battery PerformanceMarch 31, 2026Lithium iron phosphate batteries and ternary lithium batteries in lithium batteries have the advantages of high energy density, wide operating temperature range, long cycle life, and safety and reliab...view Safety First: A Multidimensional Strategy Analysis for Enhancing Lithium-ion Battery SafetyMarch 31, 2026Application and Safety Issues of Lithium-ion Batteries: Lithium-ion batteries are widely used due to their high energy density, high output power, and high average output voltage. However, accidents ...view
Safety First: A Multidimensional Strategy Analysis for Enhancing Lithium-ion Battery SafetyMarch 31, 2026Application and Safety Issues of Lithium-ion Batteries: Lithium-ion batteries are widely used due to their high energy density, high output power, and high average output voltage. However, accidents ...view